This webinar goes beyond the farm gate and explores how manure and effluent nutrients are handled for some less-than-typical farm-raised animal species. This presentation was originally broadcast on January 17, 2020. More… Continue reading “Managing manure beyond the farmyard: Manure management for less-typical species and settings”
Making Sense of Smells – Communicating Odors to Diverse Audiences
![]() Waste to Worth home | More proceedings….
Waste to Worth home | More proceedings….
|
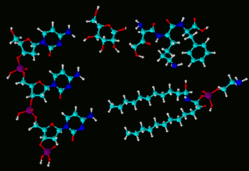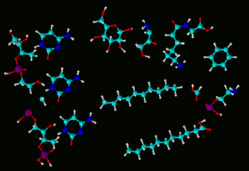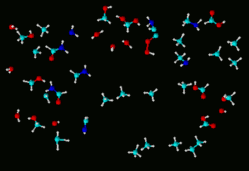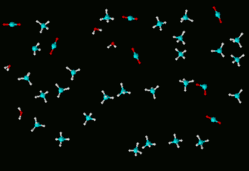
Example of odor visualization system using colors and shapes to replace chemical jargon. |
Why Is Smell and Odor Important to Animal Agriculture?
Smell is perhaps the least understood of our five senses. Yet, the human perception of odor may mean the difference between war and peace for a livestock farmer and his neighbors. Because the science of smells is complex, there is a tendency to run straight for the organic chemistry book when we try to describe farmstead odors. This approach goes right over the heads of most people. There must be a better way to communicate odors to diverse audiences. This workshop can be utilized by teachers or extension staff to teach about communication of a topic that is frequently encountered by farmers, ag professionals, and others. To see the presentation slides, scroll to the bottom of the page.
Learning Objectives
This two hour workshop will explain how to use an innovative visual technique to describe farmstead odors to general audiences without resorting to chemical jargon. The visualization technique based on shapes and colors was developed at Oklahoma State University in the mid 1990s, and has been used to talk about odors with many diverse audiences. The method demonstrates that odors have “structure”, and can be measured using the four concepts: character, concentration, intensity, and persistence.
Students will also participate in a mock laboratory exercise to demonstrate how odor intensity and pleasantness are measured. Results of the exercise will be analyzed in “real time”. Further analyses of previous exercise runs will be compared and contrasted to the workshop results. This laboratory has been presented to over 250 college freshmen and their results are presented in this recording.
Workshop Introduction
What is an Odor?
Measuring Odors
Odor Experiment
Another Odor Experiment
Physiology of Smell
 Author
Author
Douglas W. Hamilton, Associate Professor and Extension Waste Management Specialist, Oklahoma Cooperative Extension Service dhamilt@okstate.edu
Doug Hamilton is an associate professor of Biosystems and Agricultural Engineering at Oklahoma State University. He has three degrees in Agricultural Engineering from the University of Arkansas, Iowa State University, and Penn State University. His sense of smell remains keen despite the fact he has worked with livestock manure for nearly 34 years.
For More Information
The authors are solely responsible for the content of these proceedings. The technical information does not necessarily reflect the official position of the sponsoring agencies or institutions represented by planning committee members, and inclusion and distribution herein does not constitute an endorsement of views expressed by the same. Printed materials included herein are not refereed publications. Citations should appear as follows. EXAMPLE: Authors. 2013. Title of presentation. Waste to Worth: Spreading Science and Solutions. Denver, CO. April 1-5, 2013. URL of this page. Accessed on: today’s date.
Odors from Livestock Farms Curriculum Materials
One of the easiest air emissions to recognize from livestock and poultry farms is odor. It is also the most complex to characterize and study. Odors are a combination of hundreds of different emissions. Each person who smells odor interprets it differently than another person as well. With all of these variables, how can we communicate the issue of odor to students and ag professionals? These materials were developed for instructors to use in classrooms or extension programs.
Laboratory Exercise on Odor and Smell
From Dr. Doug Hamilton, Oklahoma State University
Slides and recording of author presenting the workshop. A 2 hour laboratory/workshop exercise has been presented to over 250 college freshmen.
Odor Laboratory–step by step instructions on setting up a laboratory exercise on odor
Observations and Data from Oklahoma State experience with the laboratory exercises
Video: Odors on Livestock and Poultry Farms
What role does odor play today for livestock and poultry producers? Are there ways to effectively manage odors from livestock and poultry operations and still keep the industry viable? This video examines some of the odor issues that exist in rural communities and shows examples from Nebraska of how research information is being put to use on farms.
Download a Copy of This Video
To download this video, right click on the link and select “save link as”.
Odors from Livestock Farms: A Case Study in Nebraska
File size: 34MB
Format: MP4
For More Information
Some additional resources for learning about odors and animal feeding operations:
Acknowledgements
If you have any questions or comments about the lecture or laboratory exercises, contact Dr. Doug Hamilton, Oklahoma State University dhamilt@okstate.edu. For questions on this video, contact Dr. Rick Stowell, University of Nebraska, rstowell2@unl.edu.
These materials were developed by the Air Quality Education in Animal Agriculture (AQEAA) project with with financial support from the National Research Initiative Competitive Grant 2007-55112-17856 from the USDA National Institute of Food and Agriculture.
Processing Biomass Into Biogas
Anaerobic Digestion of Animal Manures: Understanding the Basic Processes
The process of producing methane from manure is fairly straight forward – seal manure in an airtight container at a favorable temperature and it will break down into biogas: a mixture of Methane (CH4), Carbon dioxide (CO2), and trace amounts of other gases. Behind the apparent simplicity, though, lie complicated interactions involving several communities of microorganisms.
The Bioconversion Process (How Microbes Turn Organic Materials into Fuel)
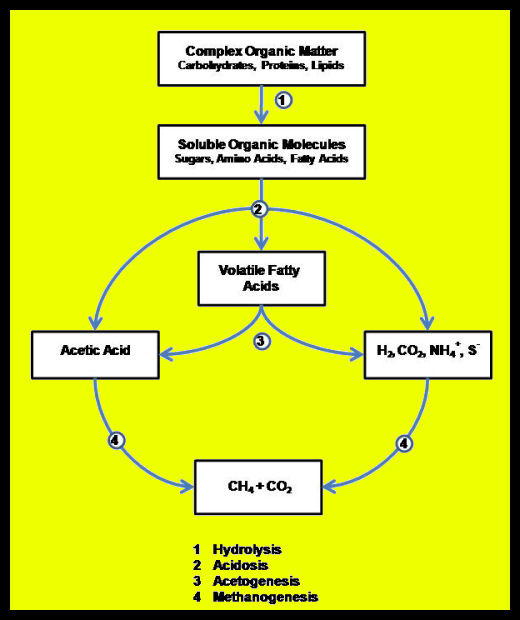
Anaerobic digestion is a multi-stage process (Figure 1) involving two to four steps, depending on where you want to draw lines in the process.
- Hydrolysis is the phase of anaerobic digestion where complex organic molecules are broken down into simpler organic molecules.
- Acidogenisis is where simple organic molecules are converted into fatty acids.
- Acetogenisis is where fatty acids are converted into acetate. This, along with acidogenisis, represents the transition from simple organic molecules to the methanogenic substrates as shown in the figure below.
- Methanogenisis is where molecules that have been converted into a suitable food source (or substrate) for methanogenic microorganisms are converted into methane by those organisms.
More than 100 different anaerobic microbes are known to contribute to the production of biogas. These microbes are organized into a number of interlinked communities. Communities of hydrolytic bacteria break complex organic matter down to simpler compounds. Acid-forming bacteria convert the simple compounds to volatile fatty acids – principally acetic acid (vinegar). Hydrolysis and acidogenisis are commonly lumped together and called anaerobic fermentation. Some microbiologists also distinguish between formation of mixed volatile fatty acids (acidosis) and reduction to Acetic acid (acetogenesis).
Methane forming Archaeans (very simple, single cell organisms similar to bacteria), called methanogens, take the end products of fermentation – volatile fatty acids, hydrogen gas (H2), CO2, and water (H2O) – and use them to form methane. Methanogens fall into two main camps depending on the pathway they use to produce methane (Figure 2). All methanogens can reduce CO2 and H2 into CH4 and H2O; those that use this pathway exclusively are called hydrotrophic methanogens. Methanogens that convert volatile fatty acids (and a number of other simple organic compounds) to CH4 and CO2 are called acetotrophic methanogens.
Limitations on Biological Processes
Anaerobic digestion is a living process, and like all biological communities, the microorganisms that carry out anaerobic fermentation and methanogensis do so under certain operating conditions.
Phases of Microbial Community Growth
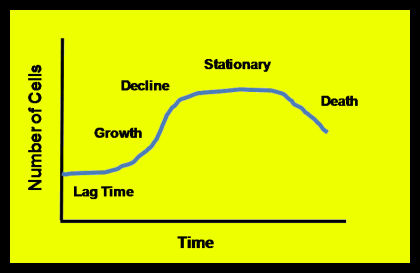
Given an ample food supply, sufficient room to expand, the absence of predators or competing organisms, all communities of organisms – be they methanogens or human beings – grow in a pattern similar to the one shown in Figure 3. The Lag Time occurs as the organisms acclimate to their environment.
- During the Growth Phase, food is not limiting, and population expands rapidly. Sometimes the Growth phase is called the Log Growth or Exponential Growth phase because the growth pattern follows an exponential curve. *Population growth slows down in the Decline Phase as the organisms meet the limit of their food supply.
- During the Stationary Phase, the community has met the limits of its food supply. Bear in mind that reproduction does not necessarily stop during the decline and stationary phases, only that the death rate approaches the reproduction rate. In some cases the community becomes dormant or goes into hibernation during the Stationary phase.
- Communities enter a Death or Endogenous Growth phase once a limited food supply is exhausted or an inhibiting element limits the further growth of organisms. During Endogenous Growth, the death rate exceeds the birth rate.
The inhibitory elements that cause endogenous growth are often the end products of a community’s metabolism.
The beauty of anaerobic digestion is that it is the work of a mixed community of organisms. The toxic end product of one community is the food supply of another. Acid forming bacteria consume the simple sugars that might inhibit hydrolytic communities. Methanogens use the acids formed in fermentation to produce CH4 and CO2. And in the end, CH4 and CO2 leave the digester as biogas.
Reproduction Time
Most anaerobic digesters are designed so that the microbial communities remain in exponential growth. An important concept to grasp is the doubling rate or reproduction time of an organism. This is the time needed for a population to double in size during exponential growth. In simple terms it is the time required for the organisms to replace themselves.
Hydraulic Retention Time
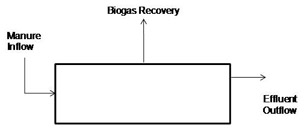
Anaerobic digestion generally takes place in a liquid, continuous flow reactor (Figure 4). If the reactor volume does not change, then flow out of the reactor equals flow into the reactor, and the average time that liquid remains in the reactor is its volume divided by the flow rate. This is called the hydraulic retention time of the reactor (HRT).
If the reactor is completely mixed, that is, the microorganisms are completely suspended in the reactor, then the time the cells remain in the reactor equals the HRT. If the HRT of a completely mixed reactor equals the reproduction time of the organisms living in the rector, a new cell is formed to replace each cell leaving the reactor, and the population within the reactor remains stable. If the HRT is shorter than the reproduction time, a new cell will not be there to replace the one leaving, and the population will decline, or “wash out.”
Solids Retention Time
Reducing HRT decreases reactor size, and smaller reactors reduce costs; therefore, many digestion systems are designed so that microorganisms remain in the reactor longer than its HRT. For instance, we could put a screen on the outlet of the reactor in Figure 4 so that some of the microorganisms are trapped inside. We can calculate cell retention time by dividing the mass of microorganisms trapped in the reactor by the mass of organisms leaving the reactor. The microbial population is kept stable by setting cell retention time equal to reproduction time.
It is easier to measure the total mass of solid particles suspended in liquid rather than the mass of viable cells; therefore, cell retention time is approximated by Solids Retention Time (SRT), or the mass of solids in the reactor divided by the mass of solids leaving.
Steady Food Supply
Microorganisms need food to reproduce and grow. Methanogens have the uncanny ability to go dormant during periods of food shortage. This is good because you can easily restart an anaerobic digester after long periods of inactivity. On the other hand, sudden increases in feeding can cause bursts of gas production, which lead to foaming and scum formation in the digester.
Temperature
Methanogens thrive in two temperature ranges. Thermophyllic (heat loving) methanogens are fast growing (reproduction time 10 to 15 days), but they operate in a fairly narrow band of temperature centered on 55 degrees C. Mesophyllic methanogens are slower growing (reproduction time up to 30 days), but they tolerate a wider range of temperatures. 35 degrees C. is optimal for mesophyllic methanogens, but they can tolerate much lower temperatures. Provided they have sufficiently long SRT, digesters operated at 20 degrees C. do not show substantial loses in gas production when compared to those operated at 35 degrees C.
Oxygen
Methanogens are strict anaerobes, meaning the least amount of oxygen is poison to them. Acid forming bacteria are more tolerant of oxygen. So, if oxygen gets into an anaerobic digester, methane concentration will drop and carbon dioxide concentration will increase in the biogas.
pH
pH is an major indicator of reactor health. A complex set of naturally occurring buffers control pH within an anaerobic digester, with organic acid-ammonia and carbonate-bicarbonate being the most important buffers. The optimum pH range for anaerobic digestion is neutral to slightly basic (pH 6.6 to 7.6).
Digester Start-up
To ensure that there is a living population of methane producing microbes in a digester, materials that contain methanogens are commonly added. When these materials come from an outside source, such as sludge from an active manure treatment lagoon or biosolids from a sewage treatment plant, it is called a “hot start.” Sometimes, digesters are brought on line with a “cold start,” meaning manure is slowly added to a liquid filled digester until gas begins to form. Hot starts are generally quicker than cold starts. Depending on digester type, a hot start can take between one and six months to bring a digester on line. It may take six months to a year for a digester to reach steady-state under a cold start.
Inhibitory Substances Commonly Found in Manure
Inhibiting elements are often called toxic, but toxicity is a misnomer. Low doses of most inhibitory agents actually stimulate biological activity. The following sets of chemicals are the major inhibitory agents found in animal manures.
Antibiotics
Antibiotics given therapeutically or fed sub-therapeutically to animals have the potential to alter the communities of microorganisms digesting manure from treated animals. The inhibitory effect of antibiotics on gas production has been shown to be minimal if high SRT is maintained and the microorganisms are given time to acclimate.
Disinfectants
Disinfectants and cleaning agents can also cause digester imbalance, although reports of upsets have not been reported in the literature. Dilution of cleaning water or bypassing the digester after cleaning is the safest tactic for handling disinfectants. Some doses may prove to have minimal effect on performance if high SRT is maintained.
Ammonia
Ammonium ion (NH4+) and its gaseous relative, Ammonia (NH3), are byproducts of protein digestion and reduction of urea. Concentrations at which Ammonical Nitrogen (NH4+NH3-N) is beneficial, inhibitory, or toxic to anaerobic digestion are given in Table 1. The toxicity of ammonia is highly dependent on pH. NH3, which is the predominant form at higher pH, is more toxic than NH4+. Usually, ammonia is not a problem in manure digesters, except for reactors treating highly nitrogenous materials such as poultry manure; and some poultry manure digesters have been able to tolerate levels as high as 6,000 mg/l if the microorganisms are given time to acclimate.
| Effect on Anaerobic Treatment | NH4+NH3-N mg/l | S– mg/l |
|---|---|---|
| Beneficial | 50-200 | <50 |
| No Adverse Effect | 200-1000 | 50-100 |
| Inhibitory at higher pH values | 1500-3000 | 100-200 |
| Toxic | >3000 | >200 |
Sulfate and Sulfide
Sulfate (SO4–) is not an inhibitory substance, per se. Its presence can reduce CH4 production because a group of bacteria called sulfate reducers can out-compete hydrotrophic methanogens for available H2. Competition with sulfate reducers does not usually reduce production from manure digesters since there is plenty of acetic acid around for acetotrophic methanogens to produce gas. The end product of sulfate reduction, Sulfide (S–), can be quite toxic to anaerobic digestion. Like ammonia, the toxicity of S– is dependent on digester pH. Stimulatory and inhibitory concentrations of sulfide are given in Table 1. Also, like ammonia, S- exists in a gaseous form, Hydrogen sulfide (H2S), so its control is a balance between source reduction, gas production, and pH.
Salt
The higher the soluble salt content in a digester, the harder microorganisms have to work in order to transport water in and out of their cells. Research has shown that it is mainly the cations (positive ions in solution) that inhibit microbial activity. Stimulatory and inhibitory concentrations of base cations are given in Table 2.
| Cation | Stimulatory (mg/l) | Moderately Inhibitory (mg/l) | Strongly Inhibitory (mg/l) |
|---|---|---|---|
| Sodium, Na+ | 100-200 | 3500-5500 | 8000 |
| Potassium, K+ | 200-400 | 2500-4500 | 12,000 |
| Calcium, Ca+ | 100-200 | 2500-4500 | 8000 |
| Magnesium, Mg+ | 75-150 | 1000-1500 | 3000 |
Precipitation of Inhibitory Substances
It should be noted that most inhibitory substances must be in solution to reduce biological activity in digesters. Therefore, the inhibitory effect of S– is reduced through precipitation of insoluble metal sulfides. Soluble Mg and NH3 are reduced by precipitation of Struvite (MgNH4PO4).
Upset Conditions and Their Control
Fermentative bacteria are generally more robust and faster growing than methanogens. The first indication that something is wrong with a digester occurs when the acid formers start to overpower the methane formers. This may show up as a drop in biogas production. First, however, the CO2 concentration in the biogas will increase, and the organic acid concentration of the reactor liquid increases. Both of these will cause a drop in pH. So, daily measurement of pH is a good method to monitor digester health. The relationship between fermentative and methanogenic communities can become so unbalanced, that even the acid formers can no longer tolerate the low pH conditions. At this point we say the digester is “stuck,” or it has “soured” or become “pickled.” Basic steps to follow in case of digester upset are:
- Reduce the feeding rate.
- Stabilize pH.
- Determine and correct the cause of the imbalance.
- Slowly increase feeding rate while maintaining neutral pH.
Sources
- Cundiff, J.S., and K.R. Mankin. 2003. Dynamics of Biological Systems. St Joseph, MI: ASABE.
- Darling, D. 2009. The Internet Encyclopedia of Science. http://www.daviddarling.info/encyclopedia/ETEmain.html. Accessed May 16, 2009
- Hamilton, D.W., P.R. Sharp, and R.J. Smith. 1985. The Operational characteristics of a manure digester for 60 beef cattle. pp 500-508, in Agricultural Waste Utilization and Management, the Proceedings of the 5th International Symposium on Agricultural Wastes. St Joseph MI: ASABE.
- McCarty, M.L. 1964. Anaerobic waste treatment fundamentals. Public Works, Sept-Dec. 1964.
- Mohr, M.A. 1983. Inhibition of methane production by ammonia nitrogen in the anaerobic digestion of poultry manure. MS Thesis. Madison, WS: University of Wisconsin.
- Ndegwa, P.M., D.W. Hamilton, J.A. Lalman, and H.J. Cumba. 2007. Effects of cycle frequency and temperature on the performance of anaerobic sequencing batch reactors treating swine waste. Bioresource Technology. 99:1972-1980
- Strauch, D. and K. Winterhalder. 1985. Effect of disinfectants, additives and antimicrobial drugs on anaerobic digestion. pp 516-522 in, Agricultural Waste Utilization and Management, the Proceedings of the 5th International Symposium on Agricultural Wastes. St Joseph MI: ASABE.
- Varel, V.H. and A.G. Hashimoto. 1982. Methane production by fermentor cultures acclimated to waste from cattle fed Monenesin, Lasalocid, Salinomycin, or Avoparcin. Applied and Environmental Microbiology. 44(6):1415.
Contributors
This document was adapted from Factsheet BAE-1747, Oklahoma State University, authored by
- Doug Hamilton, Waste Management Specialist, Oklahoma State University with contributions from:
- Daniel Ciolkosz, Extension Associate, Penn State
- Jerry Martin II, Environmental Engineer, USDA – ARS Coastal Plains Soil, Water, and Plant Research Center
- All diagrams/images cc 3.0 Doug Hamilton, Waste Management Specialist, Oklahoma State University
Peer Reviewers
- Robert Graves, Agricultural Engineer, Pennsylvania State University
- Mark Rice, Extension Specialist Waste Management, North Carolina State University
Environmental Benefits of Anaerobic Digestion
The manure handling system of any farm is made up of many different components, each with a different function and purpose. An anaerobic digester, although only one component of the system, can greatly improve the environmental performance and efficiency of the overall system. The main effect of anaerobic digestion is conversion of organic matter to biogas. This conversion has many potentially beneficial environmental and management side effects.
Odor reduction
By removing organic matter, the digester reduces the organic matter-loading and associated oxygen demand on downstream manure handling components. This may allow the downstream components to be smaller, operate more efficiently and function with less environmental impact. Anaerobic pretreatment may be a more economical method of converting an anaerobic lagoon to an aerobic lagoon, compared to mechanical aeration. Digester effluent is more stable than raw manure. It contains more stable organic material and less volatile odorants. Thus, storage and land application of digester effluent greatly reduces odor nuisance compared to raw manure.
Uses for digested solids
Manure solids are stabilized through anaerobic digestion. What was once reactive, partially digested material has been processed into stable microbial biomass and precipitated nutrients, although the majority of nutrients remain with the liquid. The potential to dry and transport digester solids is greatly improved over raw manure. The solids can be recycled and used for bedding or a soil amendment on the farm. The reduction in moisture content also increases the feasibility of selling the solids to farms that are greater distances away. In the right market conditions, composting the digested solids can result in a value-added product that can be sold to homeowners, gardeners or the landscape industry.
Plant nutrients
Plant nutrients are conserved and transformed during anaerobic digestion. Ammonium is created from manure proteins. This can be a benefit or a nuisance. If injected immediately into the soil, ammonium-rich effluent is highly available for plant growth. On the other hand, if digester effluent is stored under anaerobic conditions, ammonium will convert to ammonia gas and escape to the atmosphere. Since digesters are also a reducing environment, the potential exists for capture of ammonium and soluble phosphorus through precipitation as struvite.
Many metals are precipitated during anaerobic digestion. Sulfur is reduced to sulfide, which is generally a bad thing since it can escape as hydrogen sulfide gas. However, the digester environment can be manipulated so that sulfides are precipitated along with potentially harmful metals such as Ni and Zn.
Greenhouse gases
Anaerobic digestion results in the reduced emission of greenhouse gases. This may seem ironic, since the methane contained in the resulting biogas is a powerful greenhouse gas. An anaerobic digester is a controlled environment that captures the methane. After capture, it is either flared or used to generate electricity and/or heat.
When flared, the carbon dioxide formed in the combustion has less heat trapping potential than the original methane, and it is essentially recycled atmospheric carbon. What is released to the atmosphere through combustion of methane was once plant material formed through photosynthesis from atmospheric carbon dioxide.
When used for energy generation, the biogas replaces power that might have otherwise been created through conversion of fossil fuel. Regardless, if the biogas is flared or used for energy generation, the farmer may be eligible for carbon credit payments.
Anaerobic Digestion on Farms
With all of the potential benefits, one might wonder why relatively few farms utilize these systems. One major reason is that anaerobic digesters are expensive to install and operate. The economic benefits have, in the past, been limited to a reduction in electricity purchased by the farm, which is not enough to offset the costs of the system.
As the interest in renewable energy sources increases, farms are increasingly able to apply and receive carbon credits. Some farms also accept off-farm waste, collecting tipping fees, to co-digest with manure. In many states, more favorable net-metering laws have also made the economics more favorable. Power generated by the digester is valued at retail costs rather than wholesale costs.
The decision to install a digester is often driven by additional considerations, such as nuisance issues. A digester greatly reduces the odor potential of the manure, which also greatly reduces neighbors’ complaints and the potential for lawsuits.
At the current time, anaerobic digestion is slowly but surely increasing as a manure treatment method in the United States. Additional information is available at: Economics of Anaerobic Digesters for Processing Animal Manure.
Contributors To This Document
Author: Doug Hamilton, Oklahoma State University Waste Management Specialist
Contributors: Jill Heemstra, University of Nebraska
Reviewers: Mark Rice, North Carolina State University and Karl Vandevender, University of Arkansas
Types of Anaerobic Digesters
| Passive Systems Low Rate Systems High Rate Systems Contributors To This Article |
All anaerobic digesters perform the same basic function. They hold manure in the absence of oxygen and maintain the proper conditions for methane forming microorganisms to grow. There is a wide variety of anaerobic digesters, each performing this basic function in a subtly different way. Seven of the most common digesters are described in this article. Construction and material handling techniques can vary greatly within the main categories.
For clarity, we can divide digesters into three categories:
- Passive Systems: Biogas recovery is added to an existing treatment component.
- Low Rate Systems: Manure flowing through the digester is the main source of methane-forming microorganisms.
- High Rate Systems: Methane-forming microorganisms are trapped in the digester to increase efficiency.
Passive Systems
Covered lagoon
This system takes advantage of the low maintenance requirement of a lagoon while capturing biogas under an impermeable cover (Figure 1). The first cell of a two-cell lagoon is covered, and the second cell is uncovered (Figure 2). Both cells are needed for the system to operate efficiently. A lagoon is a storage as well as a treatment system; the liquid level on the second cell must rise and fall to create storage, while the level on the first cell remains constant to promote manure breakdown.
Since they are not heated, the temperature of covered lagoons follows seasonal patterns. Methane production drops when lagoon temperatures dip below 20 degrees C. A covered lagoon located in the tropics will produce gas year-round, but gas production will drop considerably during the winter farther north. Since sludge is stored in lagoons for up to 20 years, methane-forming microorganisms also remain in the covered lagoon for up to 20 years. This means that much of the fertilizer nutrients, particularly phosphorus, also remain trapped in the covered lagoon for a long time. If lagoon effluent is recycled to remove manure from buildings, liquid retention time is generally 30 to 60 days – depending on the size and age of the lagoon.
Low Rate Systems
Complete Mix Digester
A complete mix digester (Figure 3) is basically a tank in which manure is heated and mixed with an active mass of microorganisms (Figure 4.). Incoming liquid displaces volume in the digester, and an equal amount of liquid flows out. Methane forming microorganisms flow out of the digester with the displaced liquid. Biogas production is maintained by adjusting volume so that liquids remain in the digester for 20 to 30 days. Retention times can be shorter for thermophyllic systems. The digester can be continuously or intermittently mixed. Intermittent mixing means the tank is stirred during feeding and only occasionally between feedings. Sometimes the process takes place in more than one tank. For instance, acid formers can break down manure in one tank, and then methane formers convert organic acids to biogas in a second tank. Complete mix digesters work best when manure contains 3 percent to 6 percent solids. Digester size can be an issue at lower solids concentrations. Lower solids mean greater volume, which means you need a larger digester to retain the microbes in the digester for 20 to 30 days.
Plug Flow Digester
The idea behind a plug flow digester (Figure 5) is the same as a complete mix digester – manure flowing into the digester displaces digester volume, and an equal amount of material flows out (Figure 6). However, the contents of a plug flow digester manure are thick enough to keep particles from settling to the bottom. Very little mixing occurs, so manure moves through the digester as a plug – hence the name “plug flow.” Plug flow digesters do not require mechanical mixing. Total solids (TS) content of manure should be at least 10 or 15 percent, and some operators recommend feeding manure with solids as high as 20 percent. This means you may need to add extra material to manure to use a plug flow digester. This is not always a bad thing if you consider the added material may also be biodegradable. More degradable material means more biogas. Plug flow digesters are usually five times longer than they are wide. Recommended retention time is 15 to 20 days.
High Rate Systems
Solids Recycling
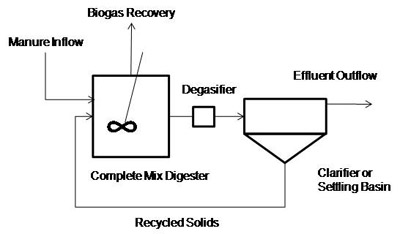
Returning some of the active organisms to the digester decreases digestion time. This is done in plug flow systems by pumping some of the effluent leaving the digester to the front of the digester. In complete mix systems, solids are settled in an external clarifier, and the microbe-rich slurry is recycled back to the digester. The systems are called contact stabilization digesters or anaerobic contact digesters (Figure 7).
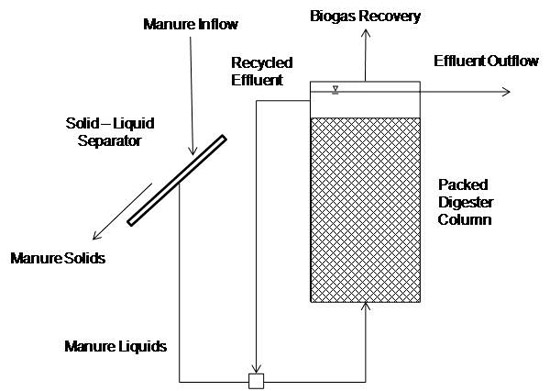
Fixed Film Digester
A fixed film digester (Figure 8) is essentially a column packed with media, such as wood chips or small plastic rings. Methane-forming microorganisms grow on the media. Manure liquids pass through the media (Figure 9). These digesters are also called attached growth digesters or anaerobic filters. The slimy growth coating the media is called a biofilm. Retention times of fixed film digesters can be less than five days, making for relatively small digesters. Usually, effluent is recycled to maintain a constant upward flow. One drawback to fixed film digesters is that manure solids can plug the media. A solid separator is needed to remove particles from the manure before feeding the digester. Efficiency of the system depends on the efficiency of the solid separator; therefore, influent manure concentration should be adjusted to maximize separator performance, usually 1 percent to 5 percent total solids). Some potential biogas is lost due to removing manure solids.
Suspended Media Digesters
In these types of digesters, microbes are suspended in a constant upward flow of liquid. Flow is adjusted to allow smaller particles to wash out, while allowing larger ones to remain in the digester. Microorganisms form biofilms around the larger particles, and methane formers stay in the digester. Effluent is sometimes recycled to provide steady upward flow. Some designs incorporate an artificial media such as sand for microbes to form a biofilm; these are called fluidized bed digesters.
Suspended media digesters that rely on manure particles to provide attachment surfaces come in many variations. Two common types of suspended media digesters are the upflow anaerobic sludge blanket digester, or UASB digester (Figure 10), and the induced blanket reactor, or IBR digester (Figures 11 and 12). The main difference between these two systems is that UASB digesters are better suited for dilute waste streams (<3-percent total suspended solids); whereas, the IBR digester works best with highly concentrated wastes (6 percent to 12 percent TS).
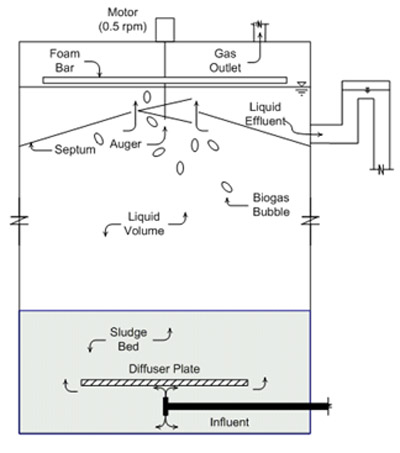
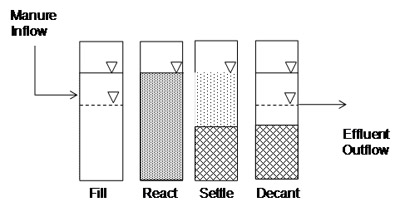
Sequencing Batch Digester
An anaerobic sequencing batch reactor (Figure 13), or ASBR digester, is a variation on an intermittently mixed digester. Methane forming microorganisms are kept in the digester by settling solids and decanting liquid. An ASBR operates in a cycle of four phases (Figure 14). The digester is fed during the fill stage, manure and microbes are mixed during the react phase, solids are settled during the settle stage, and effluent is drawn off during the decant stage. The cycle is repeated up to four times a day for nearly constant gas production. Liquid retention times can be as short as five days. Although ASBR digesters work well with manure in a wide range of solids concentrations, they are particularly well suited for very dilute manures (< 1 percent TS), and if filled with active microbes during start-up, can even produce biogas with completely soluble organic liquids. Sludge must be removed from the ASBR digester periodically. Concentrated nutrients are harvested during sludge removal.
Contributors To This Article
Author
- Doug Hamilton, Waste Management Specialist, Oklahoma State University
Peer Reviewers
- Robert Graves, Agricultural Engineer, Pennsylvania State University
- M. Charles Gould, Bioenergy Educator, Michigan State University Extension