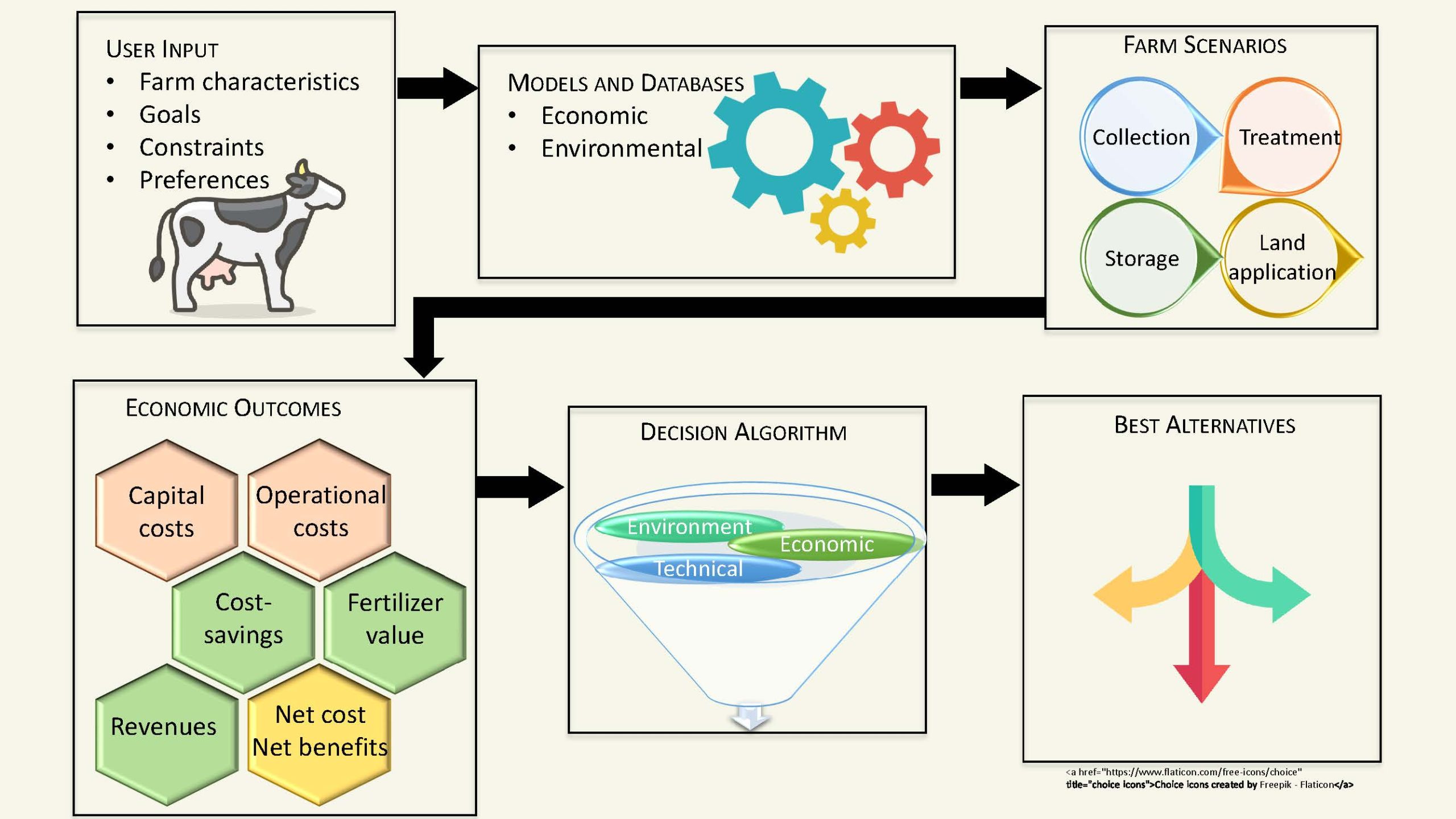
Purpose
Animal manure is frequently applied to crop fields to supplement manufactured fertilizers, as manure is rich in many of the nutrients required for plant growth. Most nutrients in manure exist in organic forms, which must first be mineralized to inorganic forms before they can be used by plants. Direct land application of manure relies on in situ mineralization of nutrients by soil microorganisms, which is a slow and difficult-to-control process. In anticipation of limited immediate nutrient availability, manure is often applied to fields in excess of actual agronomic nutrient need. The excess nutrients can leach into water sources, causing accelerated eutrophication and threatening human and ecosystem health. As such, it is advantageous to investigate technologies designed to recover manure nutrients in inorganic forms, which can then be more easily regulated and applied to suit specific agricultural demand. Bioelectrochemical systems (BES) are a novel treatment option employing electrogenic microorganisms to drive operation and recover mineralized nutrients, making them an advantageous resource recovery mechanism.
What Did We Do?
This study investigated a BES for organic nitrogen mineralization and ammonia recovery from synthetic (e.g. prepared solution of organic nitrogen and acetate) and real dairy manure. The BES was custom fabricated and followed a two-chamber design as outlined in Burns and Qin (2023). Briefly, a cation exchange membrane separated a biological anode and chemical cathode in respective chambers. Electrodes were connected via a 10 Ohm resistor to allow for current flow, and synthetic or real dairy manure was fed to the biological anode depending on the experimental condition under investigation. The BES was operated in both fuel cell and electrolysis cell (applied voltage = 0.8 V) modes.
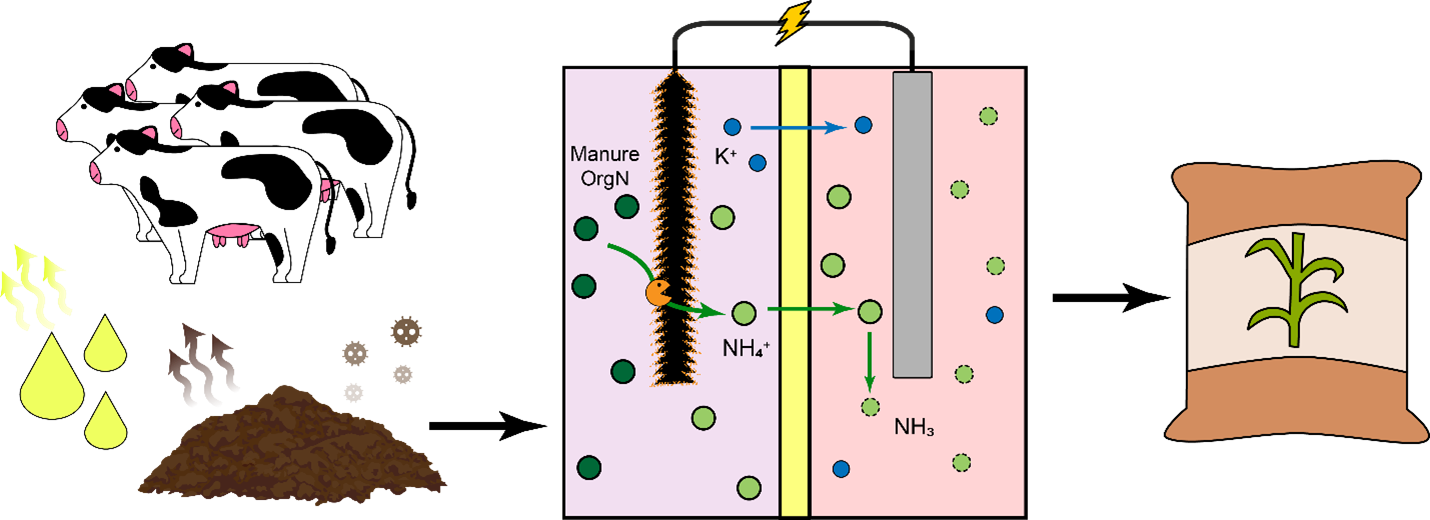
System performance was evaluated for organics removal (measured as chemical oxygen demand or COD), nitrogen removal, and total ammoniacal nitrogen production. We also calculate the nitrogen removal efficiency, RN, which measures how current is partitioned to drive nitrogen transport (as NH4+) across the cation exchange membrane. We modeled full-scale implementation of this technology on a theoretical Wisconsin dairy farm based on the experimental results obtained when treating real dairy manure. Results from the model (functional unit: tonne of manure treated, allocation: kg fat-and-protein-corrected milk (FPCM)) were used to investigate environmental impacts including greenhouse gas (GHG) emissions (in kg CO2eq tonne-1 manure), ammonia losses (in kg NH3 tonne-1 manure), and eutrophication potential (kg PO42- tonne-1 manure).
What Have We Learned?
In synthetic manure experiments, the BES consistently achieved excellent organics removal, exhibiting COD removal efficiencies well above 90%. Furthermore, total nitrogen removal efficiency averaged around 40% in electrolysis cell operation, and was seen to reach as high as 60% for some experiments. In fuel cell operation, nitrogen removal efficiency was decreased, averaging around 23%, indicating a slight advantage for nitrogen removal in electrolysis operation. RN exhibited interesting trends before, during, and after electrolysis cell operation. For the same system operational parameters, RN in fuel cell mode was around 1 mol N mol-1 electrons before electrolysis cell operation. However, during electrolysis operation and when the system returned to fuel cell operation after electrolysis cell operation, RN was and remained elevated at nearly 3 mol N mol-1 electrons with much more variability. This variability suggests that the microbial community was less tolerant to applied voltage conditions, and that there was perhaps some significant change during electrolysis operation that was difficult to recover from upon return to fuel cell operation. When treating real dairy manure, the system achieved average removals of 60% of total nitrogen and 58% of organic matter (Burns et al., 2024). The system exhibited similar nitrogen removal across multiple dairy manure feedstocks, however, a decrease in RN was observed with more complex dairy manure feedstock, likely due to the presence of competing ions (Burns et al., 2024).
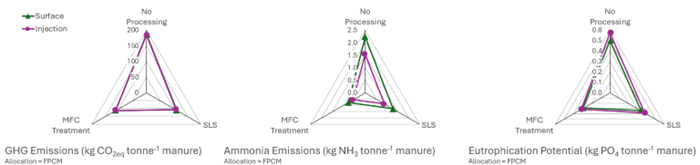
We also investigated the environmental impacts of BES manure treatment when scaled up to a ~730 cow dairy farm. Impacts on greenhouse gas emissions, ammonia losses, and eutrophication potential were compared for surface and injection application of three manure treatment scenarios: (1) no manure treatment or processing, (2) solids-liquids separation (SLS) manure processing, and (3) BES manure treatment. Preliminary results from the model reported that BES manure treatment decreased impacts in all three categories when compared to the no treatment scenarios, and resulted in less ammonia loss when compared to the SLS treatment scenarios (Figure 2). For GHG emissions, BES manure treatment had slightly increased emissions when compared to SLS, mostly due to the added energy and freshwater inputs. However, BES manure treatment received more credits for P and N-based fertilizers than SLS treatment. For eutrophication potential, BES manure treatment had slightly less impact when compared to SLS treatment, despite the added impacts of freshwater, energy, and supplemental chemicals required for the treatment. Based on these results and those from experimental data, BES manure treatment is concluded to be a promising and competitive technology worthy of further development.
Future Plans
The results of this research prove bioelectrochemical systems to be a viable manure treatment alternative to current technologies. Our future work will involve investigating the organic nitrogen degradation kinetics in the BES treating dairy manure. Our goal is to determine reaction rate orders and calculate kinetic constants for degradation of COD, TN, and organic N within the cell, which can be used to develop more accurate full-scale models of the process. This analysis can be extended to investigate differing compositions of dairy manure based on the dairy’s variable feed compositions throughout the year. Additionally, we plan to expand the environmental impact analysis to include two other comparison scenarios which would be realistic at the industrial scale: (1) minimizing freshwater inputs for manure dilution and (2) harvesting electricity produced by the BES towards meeting pumping and aeration demands. Based on the model, BES manure treatment would require approximately 1,700 kWh of electricity per week to meet pumping an aeration demands, some of which can be provided by the microbially-generated electric current in the system. Furthermore, due to reactor size constraints at the lab scale, there is currently a large amount of freshwater used to dilute the manure prior to treatment with the MFC. This work will help to contextualize BES within existing manure treatment frameworks and will help both researchers and practitioners make informed decisions regarding manure treatment options.
Authors
Presenting author
McKenzie Burns, PhD Candidate, the University of Wisconsin—Madison
Corresponding author
Dr. Mohan Qin, Assistant Professor, the University of Wisconsin—Madison, mohan.qin@wisc.edu
Additional authors
-
- Dr. Horacio Aguirre-Villegas, Scientist III, the Nelson Institute for Environmental Studies at the University of Wisconsin—Madison
- Dr. Rebecca Larson, Associate Professor, the University of Wisconsin—Madison
Additional Information
-
- Published journal articles (these are also the citations for this conference proceeding):
- Burns, M., Tang, H., Larson, R. A., & Qin, M. (2024). Bioelectrochemically-assisted ammonia recovery from dairy manure. Water Research, 252, 121243. https://doi.org/https://doi.org/10.1016/j.watres.2024.121243
- Burns, M., & Qin, M. (2023). Ammonia recovery from organic nitrogen in synthetic dairy manure with a microbial fuel cell. Chemosphere, 138388. https://doi.org/https://doi.org/10.1016/j.chemosphere.2023.138388
- Website: https://qinlab.cee.wisc.edu/
- Published journal articles (these are also the citations for this conference proceeding):
Acknowledgements
The authors would like to thank the support from National Science Foundation CBET 2219089. In addition, the authors would like to thank the startup fund from the Department of Civil and Environmental Engineering, College of Engineering, the Office of the Vice-Chancellor for Research and Graduate Education (OVCRGE) at the University of Wisconsin–Madison, and the Wisconsin Alumni Research Foundation (WARF) for the support of this study. The authors gratefully acknowledge support from Jackie Cooper of the Environmental Engineering Core Facility at the University of Wisconsin–Madison for use of facilities and equipment. Finally, the authors thank Andrew Beaudet, Ethan Napierala, Katie Mangus, and David Xiong for their contributions as undergraduate researchers on this project.
The authors are solely responsible for the content of these proceedings. The technical information does not necessarily reflect the official position of the sponsoring agencies or institutions represented by planning committee members, and inclusion and distribution herein does not constitute an endorsement of views expressed by the same. Printed materials included herein are not refereed publications. Citations should appear as follows. EXAMPLE: Authors. 2025. Title of presentation. Waste to Worth. Boise, ID. April 7–11, 2025. URL of this page. Accessed on: today’s date.