Purpose
Intensive animal husbandry produces large volumes of liquid manure with significant amounts of phosphorus, ammonium, and potassium as they pass through the feed of farm animals. As a result, direct land application of manure, the current common approach, causes environmental concerns such as soil over-fertilization and groundwater and surface water contamination, which leads to eutrophication. Manure nutrient management is, therefore, necessary to address these problems. While most engineering options are focused on phosphorus and ammonium recovery, few studies have pursued recovery methods for potassium. In this talk, we present an electrochemical technology using a sacrificial magnesium anode and a stainless-steel cathode for simultaneous recovery of phosphorus and potassium in the form of potassium-magnesium-phosphate (KMgPO4·xH2O, K-struvite).
Mg2+ + K+ + HnPO4n-3 + 6H2O = KMgPO4*6H2O + nH+
K-struvite has the potential to be used as a slow-release fertilizer and this technology will add flexibility to the manure management strategies currently available by diversifying the recoverable by-products.
What Did We Do?
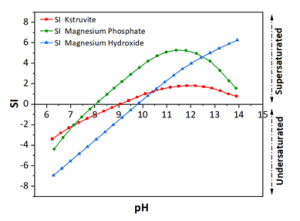
To predict the thermodynamic stability of K-struvite, a thermodynamic model was developed based on the average ion concentrations of phosphorus, and potassium measured in real liquid pig manure (Figure 1). According to this model, magnesium phosphate is a possible by-product of K-struvite precipitation. Also, the probable formation of magnesium hydroxide was enhanced with increasing pH value due to the increase in hydroxide ion concentration. As a result, the ideal range for precipitation of K-struvite lies at pH values between 10 and 11.
To understand the role of pH on K-struvite formation, a 50 mM KH2PO4 solution was used to perform the preliminary batch electrochemical experiments. A constant voltage of -0.8 V vs. the Ag/AgCl reference electrode was applied to the pure magnesium anode using a potentiostat. One experiment was performed on the natural pH of the initial solution, 4.5, while potassium hydroxide was used to raise the initial pH of the second experiment to 9.5.
What Have We Learned?
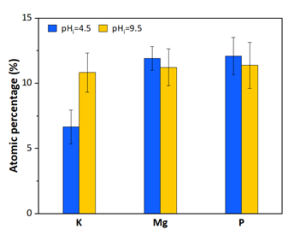
Energy-dispersive x-ray spectroscopy (EDS) of the recovered precipitates (Figure 2) indicate that by raising the initial pH from 4.5 to 9.5 the amount of potassium is increased in the precipitates. Also, due to the equimolar ratios of K:Mg:P at pH=9.5, the produced precipitates are likely K-struvite, while the pH= 4.5 sample likely contains some amount of magnesium phosphate.
This process also eliminates the disadvantages of the commonly used chemical precipitation methods, including magnesium salt dosing, and adding base to the system for pH control, due to in situ magnesium corrosion and hydroxide production at the magnesium anode surface. These advantages could potentially reduce the operating cost of the system and eliminate the addition of unnecessary salinity to wastewater through magnesium salt dosing.
Future Plans
Further investigation by using multiple characterization techniques (e.g., x-ray diffraction (XRD) and Fourier transform infrared spectroscopy (FT-IR)) is necessary to identify the exact nature of precipitates. The initial experiments will be repeated at additional pH values to further understand the role of pH on the precipitation of K-struvite in the simplified synthetic wastewater and to further detail the characterization of the composition and morphology of K-struvite precipitates. These experiments are valuable , particularly because there are few literature reports that detail the physical and chemical structure of K-struvite.
Authors
Presenting author
Amir Akbari, Ph.D. Candidate, Department of Chemical and Biomedical Engineering, Pennsylvania State University
Corresponding author
Lauren F. Greenlee, Associate Professor, Department of Chemical and Biomedical Engineering, Pennsylvania State University
Corresponding author email address
greenlee@psu.edu
Additional Information
Once completed, future publications and data repository information will be available at https://sites.psu.edu/greenlee/
Acknowledgements
The authors would like to thank the U.S. Department of Agriculture, NIFA AFRI Water for Food Production Systems (#2018-68011-28691) for providing the funding support of this research through the “Water and Nutrient Recycling: A Decision Tool and Synergistic Innovative Technology” project.
The authors are solely responsible for the content of these proceedings. The technical information does not necessarily reflect the official position of the sponsoring agencies or institutions represented by planning committee members, and inclusion and distribution herein does not constitute an endorsement of views expressed by the same. Printed materials included herein are not refereed publications. Citations should appear as follows. EXAMPLE: Authors. 2022. Title of presentation. Waste to Worth. Oregon, OH. April 18-22, 2022. URL of this page. Accessed on: today’s date.

