Proceedings Home | W2W Home 
Purpose
Greenhouse gas (GHG) emissions from dairy manure can be affected by barns, bedding and manure collection, as well as processing and storage. To reduce life cycle environmental impacts of milk production, it is important to understand the mechanisms involved in production and emission of GHGs from dairy manure. In addition to the GHGs emitted from the manure surface, the production of these gases in manure at different depths is an important but poorly understood driver of emissions. Because it is often not practical to measure GHG production and emissions directly in the field, simulation of these processes, both experimentally and through modeling, is needed to help understand the GHG emission mechanisms.Because manure samples are heterogeneous and their composition varies based on the bedding materials and bedding rate as well as cleaning frequency, it is also necessary to consider the impacts of these different types of manure heterogeneity and their impact on emission processes. Another important element that can impact GHGs emissions from dairy manure is oxygen. GHG emission rates can be different based on manure storage status (aerobic, anaerobic, and mixed conditions) and storage time. Several other factors, such as manure bedding materials, bedding rate, applied stress, temperature and moisture content can also impact the microbial activities that produces these GHGs. Our goals are to enhance understanding of the relationships between these factors and GHG emissions from dairy manure, and to identify strategies by which substantial reductions in GHG can be realized in a practical way.
What did we do?
In a controlled laboratory environment we investigated three different dairy manures: sand stacked manure, sawdust bedded manure, and organic sawdust bedded manure. The first two manures were studied and measured in 2016, and the last one was collected and measured in February 2017. After sample collection, manures were mixed in a cement blender to be more homogeneous, and were then transported to buckets and jars for compaction and storage. Nine buckets were filled with manure in layers, and each layer was characterized for physical and biochemical properties. Three levels of stress (0 N/m2, 4196 N/m2, and 12589 N/m2) were applied above the manure to emulate the impact of overburden at various pile depths. Manure bulk density and permeability for each bucket were measured, and the average of each treatment was summarized to evaluate relationships with GHG emissions. Four gases (NH3, CH4, CO2, and N2O) were investigated. The manure moisture content and water holding capacity were measured adjusted to create aerobic, anaerobic, and mixed conditions for manure microorganisms. Three moisture contents were applied to 300 g manure samples, each three replicates. Each manure storage condition was simulated in 2L glass vessels for five durations (one day, two weeks, one month, two month, and three months). The relationship between storage time and GHG rates was assessed.



What have we learned?
The results showed that there are good prospects that GHGs reductions can be realized in dairy manure management. In this work, manure that was characterized between each sample layer in the buckets showed similar results, which means the samples are pretty homogeneous. Bulk density and permeability decreased with increasing applied stress. GHG emissions and ammonia emissions showed correlation with the compaction density. Using different bedding materials did impact the GHGs rate.
Future Plans
The combination of prediction models (DNDC and IFSM) and real-word data will be discussed next.
Corresponding author, title, and affiliation
Fangle Chang, post-doctoral at Penn State University, State College PA
Corresponding author email
Other authors
Micheal Hile, Eileen E. Fabian (Wheeler),
Additional information
Micheal Hile, mlh144@psu.edu
Eileen E. Fabian (Wheeler), Professor of Agricultural Engineering, Environmental Biophysics, Animal Welfare, and Agricultural Emissions, Integrated Research and Extension Programs, Penn State University, State College PA, fabian@psu.edu
Tom L. Richard, Professor of Agricultural and Biological Engineering, Director of Penn State Institutes for Energy and the Environment, Bioenergy and Bioresource Engineering, Penn State University, State College PA, tlr20@psu.edu
Acknowledgements
This material is based upon work that is supported by the National Institute of Food and Agriculture, U.S. Department of Agriculture, under award number 2013-68002-20525. Any opinions, findings, conclusions or recommendations expressed in this publication are those of the author(s) and do not necessarily reflect the view of the U.S. Department of Agriculture.
The authors are solely responsible for the content of these proceedings. The technical information does not necessarily reflect the official position of the sponsoring agencies or institutions represented by planning committee members, and inclusion and distribution herein does not constitute an endorsement of views expressed by the same. Printed materials included herein are not refereed publications. Citations should appear as follows. EXAMPLE: Authors. 2017. Title of presentation. Waste to Worth: Spreading Science and Solutions. Cary, NC. April 18-21, 2017. URL of this page. Accessed on: today’s date.








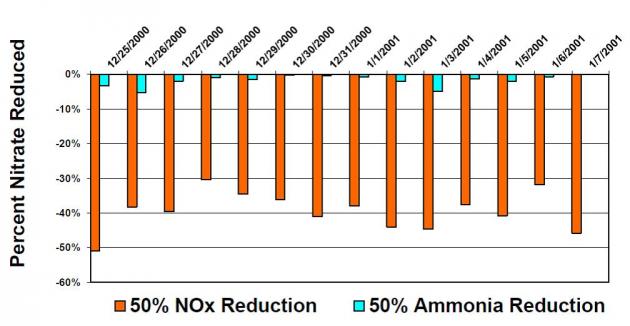 Atmospheric modeling has demonstrated that controlling NOx is the most effective strategy to reduce ammonium nitrate concentrations in the San Joaquin Valley and controlling ammonia has little effect on these concentrations. The California Air Resources Board conducted multiple modeling runs to simulate the formation of PM2.5 in the San Joaquin Valley and compare the effect of reducing various pollutants on PM2.5 concentrations. As seen in Figure 2, U.S. EPA’s Community Multi-scale Air Quality (CMAQ) indicated that reducing NOx by 50% reduced nitrate concentrations by 30% to 50% reductions, while reducing ammonia by 50% resulted in less than 5% reductions in nitrate concentrations. Similarly, the UCD/CIT photochemical transport model indicated that for the conditions on January 4-6, 1996 in the San Joaquin Valley, controlling NOx emissions is far more effective for reducing nitrate concentrations than controlling ammonia.
Atmospheric modeling has demonstrated that controlling NOx is the most effective strategy to reduce ammonium nitrate concentrations in the San Joaquin Valley and controlling ammonia has little effect on these concentrations. The California Air Resources Board conducted multiple modeling runs to simulate the formation of PM2.5 in the San Joaquin Valley and compare the effect of reducing various pollutants on PM2.5 concentrations. As seen in Figure 2, U.S. EPA’s Community Multi-scale Air Quality (CMAQ) indicated that reducing NOx by 50% reduced nitrate concentrations by 30% to 50% reductions, while reducing ammonia by 50% resulted in less than 5% reductions in nitrate concentrations. Similarly, the UCD/CIT photochemical transport model indicated that for the conditions on January 4-6, 1996 in the San Joaquin Valley, controlling NOx emissions is far more effective for reducing nitrate concentrations than controlling ammonia.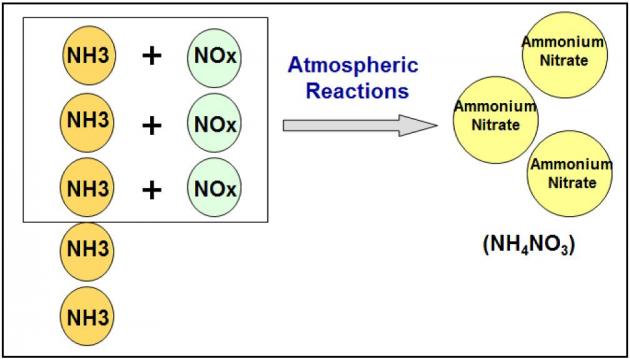 Ammonium nitrate particulate is limited by NOx in the San Joaquin Valley
Ammonium nitrate particulate is limited by NOx in the San Joaquin Valley As explained in detail in the San Joaquin Valley Air Pollution Control District 2012 PM2.5 Plan, the best scientific information available indicates that controlling NOx emissions is the most effective strategy to reduce secondary ammonium nitrate in the San Joaquin Valley. While ammonia has been demonstrated to not significantly contribute to PM2.5 concentrations in the San Joaquin Valley, the District has developed control strategies, via stringent regulations (Confined Animal Facilities – Rule 4570, Organic Material Composting – Rule 4566, Biosolids, Animal Manure, and Poultry Litter Operations – Rule 4565), that have resulted in significant reductions in ammonia emissions.
As explained in detail in the San Joaquin Valley Air Pollution Control District 2012 PM2.5 Plan, the best scientific information available indicates that controlling NOx emissions is the most effective strategy to reduce secondary ammonium nitrate in the San Joaquin Valley. While ammonia has been demonstrated to not significantly contribute to PM2.5 concentrations in the San Joaquin Valley, the District has developed control strategies, via stringent regulations (Confined Animal Facilities – Rule 4570, Organic Material Composting – Rule 4566, Biosolids, Animal Manure, and Poultry Litter Operations – Rule 4565), that have resulted in significant reductions in ammonia emissions.