Microbes are abundant in manure and food waste. Harnessing and/or controlling microbial action in a treatment process is challenging – but some technologies are making headway. This webinar will present pilot-scale research for manure and food waste treatment. This presentation was originally broadcast on March 21, 2025. Continue reading “Mobilizing Microbes in Treatment Processes”
Methane leakage imaging, detection, and quantification from dairy manure biogas capture systems
Purpose
One of the key reasons to implement manure anaerobic digestion (AD) to energy or an impermeable cover and flare (CF) system is to reduce greenhouse gas (GHG) emissions, especially methane (CH4), a potent GHG that makes up most of the US agricultural footprint. These systems that process or store manure, commonly liquid dairy or swine manure, are often referred to as biogas capture systems because they keep oxygen out and contain the manure gases that form primarily from the breakdown of organic matter by microorganisms. The biogas captured is then directed through collection pipes to a utilization system, where the goal is to convert the methane to the less potent carbon dioxide (CO2) via either combustion or electrochemical conversion. For AD systems, the biogas collected is consistent enough to burn or convert for useful energy. For CF systems, particularly those used in the Northeast and Upper Midwest, the biogas collected under the liquid manure storage cover is highly variable throughout the day and year, making it more suitable to flare the methane in the biogas rather than harvest energy. Biogas capture systems must be operated and maintained to avoid methane leaks and venting, particularly to realize their carbon reduction value that can often be monetized. Tools to easily identify point-source biogas losses, such as an optical gas imaging (OGI) camera, are still relatively costly for a bioenergy operation, however they can be used to periodically survey and conduct find it and fix it campaigns to repair and correct problems that may have gone unseen to the naked eye. The ability to better understand where and how biogas leaks and vents occur in AD and CF systems enables better design, operation, maintenance, and public confidence.
What Did We Do?
Twelve biogas capture systems operating on commercial dairy farms in NYS were surveyed once per quarter for at least a year for point-source methane losses using an optical gas imaging (OGI) camera (Teledyne FLIR GF77 uncooled) tuned to the infrared spectrum wavelength range (7 – 8.5 micrometers) where methane gas is absorbed. Any methane loss visualized with the OGI camera was recorded and its characteristics described and reported back to the farm or system owner. Other observations about the methane loss were recorded and losses were measured and/or quantified when feasible. The apparent size of the biogas loss was recorded, primarily by distinguishing between OGI visibility in “normal” camera mode versus “high sensitivity mode (HSM)”. Unique losses versus repeated (by visit) were tracked, indicating ease and motivation to correct the loss. Biogas vents were distinguished from biogas leaks, by characterizing a leak as an unknown or unintended biogas loss during normal operation. Biogas venting was considered loss that occurred by design during abnormal operating conditions, such as overpressure in the digester vessel that could not be immediately corrected with flaring excess biogas.
What Have We Learned?
This work is continuing through this year, and eight sites are completed so far. The results from those sites, that include four AD to energy systems (three electricity generation and one biomethane production) and four CF dairy manure storage systems, have generally highlighted that AD systems experience biogas venting more than biogas leaking whereas CF systems experience more leaking than venting. The number of unique biogas losses found was higher in CF systems than in AD systems, which may be due to their much larger biogas capture surface area that is also susceptible to damage from wind, wildlife, and thermal stress. Additionally, the biogas collection and flare struggle with variable biogas flow, quality, and operational robustness that results in lack of combustion during prolonged periods of the year. Another observation, which requires additional data collection from AD to biomethane systems to have confidence in, is that AD to electricity systems can result in biogas venting and/or unnoticed leaking when the biogas produced is greater than what the installed electric capacity can utilize. Additionally, most if not all AD to biomethane systems are instrumented to detect and measure biogas losses as part of their verification requirements for carbon market programs, making it less likely for losses to go unnoticed or unaddressed.
Future Plans
A methane loss detection protocol for both AD to energy systems and CF manure storage systems was developed by Cornell CALS PRO-DAIRY that has been improved during this project and will continue to evolve. Once the full 12 sites are completed, the protocol will be shared more broadly for reference, and best practices recommended for operations and maintenance to prevent, find, and correct biogas losses. Follow on work may include additional methane loss detection with total loss measurement of AD vessels and manure storage covers, to verify assumed loss rates used as defaults in GHG accounting.
Authors
Presenting & corresponding author
Lauren Ray, Sr. Extension Associate, Cornell University – PRO-DAIRY, LER25@cornell.edu
Additional authors
Jason P. Oliver, Dairy Environmental Systems Engineer, Cornell University PRO-DAIRY;
Peter Wright, Agricultural Engineer, Cornell University
Additional Information
https://cals.cornell.edu/pro-dairy/our-expertise/environmental-systems/climate-environment
Acknowledgements
This work is sponsored by the New York State Department of Agriculture and Markets. Special thanks to our collaborating dairy farms and biogas capture system operators.
The authors are solely responsible for the content of these proceedings. The technical information does not necessarily reflect the official position of the sponsoring agencies or institutions represented by planning committee members, and inclusion and distribution herein does not constitute an endorsement of views expressed by the same. Printed materials included herein are not refereed publications. Citations should appear as follows. EXAMPLE: Authors. 2025. Title of presentation. Waste to Worth. Boise, ID. April 7-11, 2025. URL of this page. Accessed on: today’s date.
Phosphorus Recycling from Dairy Manure via Hydrochar – Experience from the Lab-Scale to Pilot-Scale Hydrothermal Carbonization Prototype
Due to a technical glitch, the beginning of the recorded presentation was not recorded. Please accept our apologies.
Purpose
To address the depletion of phosphorus resources and the environmental issues associated with phosphorus enriched runoff from the application of raw manure, a strategic and sustainable approach is to recycle phosphorus from dairy manure using innovative and efficient methods. Hydrothermal carbonization (HTC) has been considered one of the sustainable techniques, which can transform dairy manure into phosphorus-enriched hydrochar at relatively low temperatures, typically ranging from 180 to 250 °C. This process not only recovers valuable phosphorus but also converts organic waste into a stable, nutrient-rich product that can be used as a soil amendment or phosphorus fertilizer.
Despite the massive experimental activity performed to characterize the HTC process, the design and development of a validated bench-scale model is crucial for scaling up. While numerous studies have explored the HTC process at the laboratory level, only a limited number of studies have assessed the technical feasibility and performance of implementing this process on an industrial scale. In this context, the purpose of this study was to provide a detailed and systematic examination of phosphorus recovery from dairy manure using a lab-scale HTC reactor and illustrated the basis of the design of a bench-scale processor and evaluated its performance in terms of hydrochar yield (HY) and phosphorus recovery (PR).
What Did We Do?

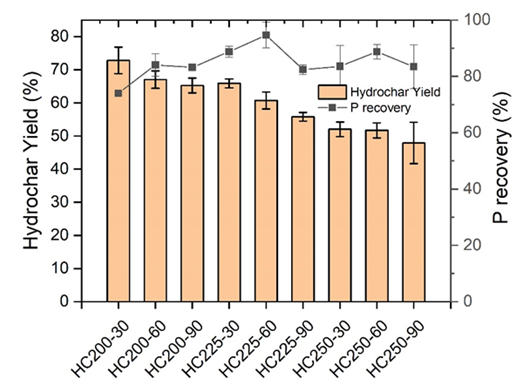
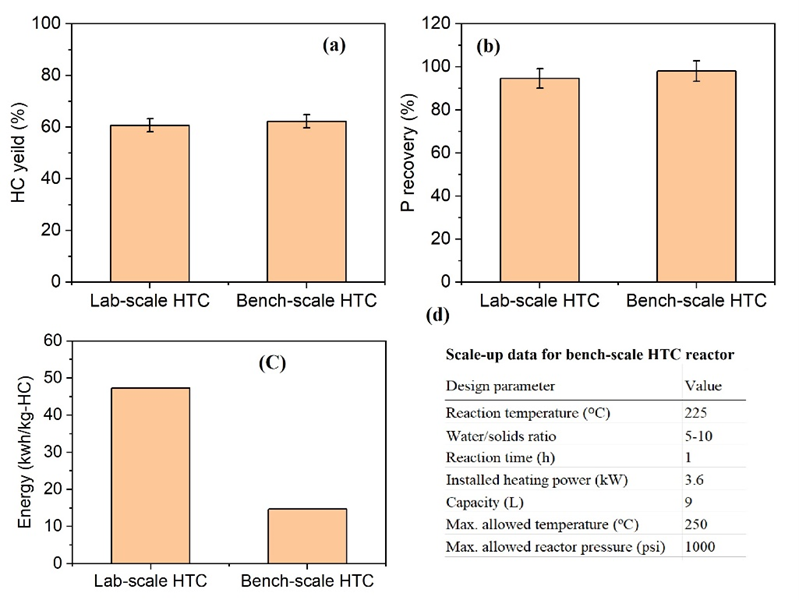
In this study, the HTC of raw dairy manure with a 7.9% of total solids was first conducted using the lab-scale reactor to optimize the process parameters, including temperature and reaction time, and then scaled up at a scale of 30 times larger (Fig. 1). The HTC-derived hydrochar samples were named according to the temperature and reaction time. For example, HC200-30 represents the hydrochar sample obtained at 200 °C and 30 min. The scaled-up reactor was designed and operated at the optimized conditions obtained from the lab-scale study, which was 225 ºC and 60 min of reaction time. The HY, which also reflects the mass reduction of dairy manure (on a dry weight basis), and PR were the two main parameters evaluating the HTC of dairy manure. We additionally evaluated the energy required for hydrochar processing in both lab- and bench-scale processors.
The HY and PR expressed as a percentage were determined by the following equations:

What Have We Learned?
Fig. 2 shows the effects of HTC processing temperature and reaction time on the HY and PR using the lab-scale reactor. It was observed that the HY decreased gradually as the processing temperature and time increased, which is attributed to the temperature and time dependent degradation of organic matter during HTC. The highest PR was observed at 225 ºC and 60 min.
As shown in Fig. 3, the scale-up of the hydrothermal carbonization (HTC) process demonstrated that HY and PR remained consistent between lab-scale and bench-scale systems, indicating that the transition to a larger reactor did not compromise HY or PR. Notably, the energy input per mass of hydrochar was significantly reduced in the bench-scale system, improving overall energy efficiency. These findings indicate that scaling up HTC can enhance process feasibility while maintaining similar nutrient recovery.
Future Plans
We plan to further develop a continuous-flow HTC system at pilot-scale as a potential advanced manure processing pathway. We will also conduct technoeconomic and environmental assessments to verify scalability and sustainability.
Authors
Presenting author
Mohammad Nazrul Islam, Postdoctoral Fellow, University of Idaho
Corresponding author
Lide Chen, Professor, Dept. of Soil & Water Systems, University of Idaho, lchen@uidaho.edu
Additional author
Brian He, Professor, Dept. of Chemical and Biological Engineering, University of Idaho
Acknowledgements
This work is supported partially by USDA NIFA (award number 2021-67022-35504) and the University of Idaho P3R1 grant.
The authors are solely responsible for the content of these proceedings. The technical information does not necessarily reflect the official position of the sponsoring agencies or institutions represented by planning committee members, and inclusion and distribution herein does not constitute an endorsement of views expressed by the same. Printed materials included herein are not refereed publications. Citations should appear as follows. EXAMPLE: Authors. 2025. Title of presentation. Waste to Worth. Boise, ID. April 7-11, 2025. URL of this page. Accessed on: today’s date.
Ammonia Recovery from Anaerobically Digested Dairy Manure Using Electrodialysis Coupled with A Hydrophobic Gas-permeable Membrane for Stripping
Due to a technical glitch, the beginning of the recorded presentation was not recorded. Please accept our apologies.
Purpose
Nitrogen is considered an essential macronutrient for plant growth and development. Ammonia, a key part of the nitrogen cycle, arises through two main pathways: naturally through biological nitrogen fixation bacteria and artificially through the Haber-Bosch process. The Haber-Bosch is an energy-intensive process relying on fossil fuels and contributing to greenhouse gases emission. Recovering ammonia from anaerobically digested dairy manure offers a more sustainable alternative to this energy-intensive process, reduces reliance on fossil fuels and mitigates environmental impact. Furthermore, the recovered ammonia can be used as a value-added product to improve soil health and sustainable agricultural productivity. Various technologies have been applied to recover ammonia from dairy manure. However, these technologies were not very efficient in terms of energy consumption, resource recovery, and treatment time. The purpose of this research was to develop a hybrid system where electrodialysis and membrane stripping were applied simultaneously to enhance ammonia recovery from anaerobically digested dairy manure within a shorter treatment period. This approach promotes circular economy through transforming dairy waste into a valuable resource.
What Did We Do?
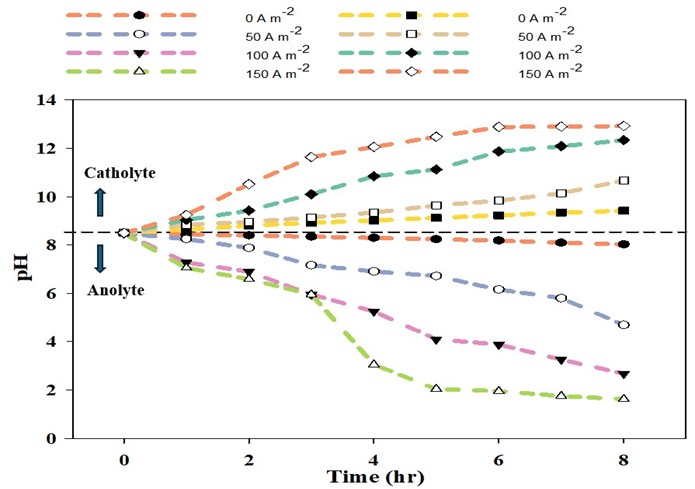
We developed a three-chamber electrodialysis and membrane stripping (ED-MS) combined system, in which the anode and the cathode chambers were separated by a cation exchange membrane (CEM), and the cathode and trap chambers were separated by a hydrophobic gas-permeable membrane (GPM). The GPM was used for membrane stripping. The authigenic acid in anolyte and the authigenic base in catholyte have been utilized as absorbents and stripping agents to improve ammonia recovery in the ED-MS system. We have applied different current densities ranging from 0 to 150 A/m2 to observe the effect of ammonia removal and recovery efficiency within an 8-hour treatment period. We also observed the maximum rate of nitrogen flux passing through the CEM and GPM for a specific energy consumption.
What Have We Learned?
From this research, we have learned that with increasing current density the removal and recovery efficiency of ammonia also increased. The presence of ammonia in the trap chamber increases with increasing treatment time (Figure 1). This ED-MS treatment process demonstrated a broad range of effectiveness for current densities ranging from 0 to 150 A/m2, achieving ammonia removal efficiencies of 42.97% to 99.49% and recovery efficiencies of 11.7% to 75.9% over an 8-hour treatment time. The main reason for this increment is to accelerate the electrolysis process and increase the rate of acid production in the anolyte and base production in the catholyte (Figure 2). The product recovered was ammonium sulfate which can be used as a fertilizer. The highest nitrogen flux through CEM and GPM was identified as 616.1 and 207.6 g-N m-2d-1, respectively, with a specific energy consumption of 45.3 kWhkg-1N-1 (Figure 3). Therefore, this research supports the idea that the ED-MS technique could be a viable solution for sustainable ammonia recovery from anaerobically digested dairy manure on a large scale.


Future Plans
To continue this research, we have a plan to investigate the reaction kinetics of this ED-MS hybrid system. Further, we will develop a novel electrochemical reactor to recover nitrogen and phosphorus simultaneously from dairy waste.
Authors
Presenting author
Ashish Kumar Das, Ph.D. Student, Environmental Science Program, College of Natural Resources, University of Idaho
Corresponding author
Dr. Lide Chen, Professor, Department of Soil and Water Systems, Twin Falls Research and Extension Center, University of Idaho, lchen@uidaho.edu
Acknowledgements
This research was funded by the USDA Sustainable Agricultural Systems Initiative through the Idaho Sustainable Agriculture Initiative for Dairy (ISAID) grant (Award No. 2020-69012-31871).
The authors are solely responsible for the content of these proceedings. The technical information does not necessarily reflect the official position of the sponsoring agencies or institutions represented by planning committee members, and inclusion and distribution herein does not constitute an endorsement of views expressed by the same. Printed materials included herein are not refereed publications. Citations should appear as follows. EXAMPLE: Authors. 2025. Title of presentation. Waste to Worth. Boise, ID. April 7-11, 2025. URL of this page. Accessed on: today’s date.
Evaluation of the aerosol transmission of the porcine reproductive and respiratory (PRRS) virus from swine production systems
Purpose
Porcine reproductive and respiratory syndrome virus (PRRSV) is a major concern to the U.S. swine industry due to the severe economic loss it can cause. Its symptoms include severe flu-like symptoms, respiratory distress, fever, and premature abortions in pregnant sows. The virus is spread during close contact between pigs or exposure to contaminated urine, semen, feces, and nasal and mammary secretions (1). Control measures have proven exceedingly costly with PRRSV which causes an estimated $1 billion in lost production in the U.S. pork industry per year (3), an 80% increase from a decade earlier (2)(4). With very few, truly effective methods available to control PRRSV after the start of an outbreak, developing methods to mitigate the dispersion of the virus has become a major priority.
Common biosecurity measures for swine operations (e.g., controlled access, personal hygiene, animal management, pest control, and production area cleaning and disinfection) have proved insufficient to stop PRRSV transmission. Producers are, therefore, seeking to understand the potential risks posed by more novel transport methods. Observations of new PRRSV cases emerging during manure handling activities have raised questions about aerosolized manure as a potential transmission vector. This study was conducted to test this possibility in the following stages:
-
- Verify the presence of viable virus sample within pit manure, lagoon samples, or dust coming from barns with active PRRSv outbreaks.
- Develop a reliable method for collecting and preserving viable airborne viral samples.
- Assess the aerosol transmission “footprint” of PRRSV originating from positive swine farms to improve understanding of potential farm-to-farm disease transmission risks.
What Did We Do?
Novel air sampling devices were constructed by the project team (Figure 1) to be deployed inside and outside swine production units to accumulate samples of particulates and aerosols. The devices accommodate a commercially available Air Prep filter cartridge (innovaprep.com) to capture particulates pulled across the filter by a fan housed within the sampling unit.


Our project team worked closely with the lead veterinarian at a large swine integrator in Nebraska to access farms within 5 to 7 d of pigs being confirmed PRRSV-positive. Sampling events 1 and 2 focused on evaluating PRRSV presence on indoor surfaces, fresh and stored manure, flies, and maggots. Sampling events 3 through 5 focused on evaluating PRRSV presence in air downwind of PRRSV-positive swine production areas or downwind of land application of manure from PRRSV-positive animals.
Sampling Event 1. A swine breeding operation was identified where animals were currently testing positive for and showing clinical signs of PRRSV infection. At this site, two production areas were selected at random for sampling. Surface swabs were collected from floors, fan louvers, and pen dividers. Fresh fecal samples were collected from sows in the same production areas, and an air sampler was placed on the floor in each room and allowed to operate for two hours before retrieving the filters. For surface samples, sterile swabs were swept over each surface type and then placed into phosphate buffered saline (PBS) elution buffer. Fresh fecal samples were collected using a sterile spatula and placed into clean sample containers. Upon retrieving filters from air samplers, a sterilized knife was used to separate the filter from the plastic casing in which it was mounted, and sterile forceps were used to transfer the filter into a PBS elution tube. All samples were transported on ice to the University of Nebraska-Lincoln (UNL) Schmidt Lab and then submitted to the Iowa State University Veterinary Diagnostic Laboratory for analysis by polymerase chain reaction (PCR).
Sampling Event 2. A swine finisher unit was identified where animals were currently testing positive for and showing clinical signs of PRRSV infection. At this site, two production areas were selected at random for sampling inside the building. Surface swabs were collected from floors, fan louvers, feeders, and pen dividers. An air sampler was placed on the floor in each room and allowed to operate for four hours before retrieving the filters. Additional air samplers were mounted outside the building. For one production area, three samplers were mounted at a height aligning with the center of a minimum ventilation fan and spaced at 5, 12, and 19 feet from the rim of the fan hood. For a second production area, two samplers were mounted at a height aligning with the center of a minimum ventilation fan and spaced at 5 and 13 feet from the rim of the fan hood. These samplers were allowed to run for three hours before filters were retrieved. For surface samples, sterile swabs were swept over each surface type and then placed into PBS elution buffer. Manure samples from two deep pit storage sections of the building were collected using a plastic pole and dipper cup and placed into clean plastic bottles. Maggots observed in one pump out port were collected by hand and placed into PBS elution buffer. Upon retrieving filters from air samplers, a sterilized knife was used to separate the filter from the plastic casing in which it was mounted, and sterile forceps were used to transfer the filter into a PBS elution tube. Flies present around the production buildings were also collected at this site. For one sample, approximately six flies were captured and placed directly into PBS elution buffer. For a second sample, approximately six flies were captured, placed into 70% EtOH for 10 s, and then transferred from the ethanol to PBS elution buffer. All samples were transported on ice to the UNL Schmidt Lab and then submitted to the Iowa State University Veterinary Diagnostic Laboratory for analysis by PCR.
Sampling Event 3. A naturally-ventilated PRRSV-positive swine farm was identified. Air samplers mounted on t-posts were deployed in an array at a height above the ground of roughly 6 ft at varying distances (10 yards to 1 mile) from the buildings after using smoke candles to confirm wind direction and dispersion. Sampling was conducted for approximately 2.5 hours on a day with 40-55°F temperature,10-20 mph winds, and full cloud cover (Figure 2).
Sampling Event 4. At a mechanically-ventilated PRRSV-positive swine farm, sampling was conducted using the same process as for Event 3 for approximately 21.25 hours starting on a day with 85-105°F temperature, 4-10 mph winds, and full sun exposure, then continuing overnight.
Sampling Event 5. Using the previously described process, sampling was conducted for approximately 2.5 hours on a day with 70-95°F temperature, 2-10 mph winds, and partly cloudy conditions downwind of a field where lagoon effluent from PRRSV-positive pigs was being applied via center pivot.

All samples were submitted to the Iowa State Veterinary Diagnostic Lab for RT-qPCR analysis to identify PRRS viral genomic material.
What Have We Learned?
Results of PCR analyses for sampling event 1 (Table 1) revealed that, in barns where swine oral fluid samples were positive for PRRSv, all surface samples collected were also positive or suspected positive for PRRSv. The same was true for all of the surface and air samples collected inside the barn and for the air samples located up to 19 ft minimum from the building ventilation fans during sampling event 2 (Table 2). Maggots taken from the manure pit during sampling event 2, along with sterilized and unsterilized flies, tested positive for PRRSV, as well. Conversely, all manure samples obtained during sampling event 2 tested negative using the methodologies employed. This outcome does not dismiss manure as a possible transmission source; rather, it underscores the need for ongoing research to develop a reliable detection method for PRRS within such a complex matrix.
The team has not yet recovered air samples testing positive for PRRSV from any of the exterior arrays in sampling events 3-5 (Table 3). This could be due to ambient air conditions during the tests which may have caused rapid destruction of the virus or dilution of the virus below detectable concentrations. The rolling terrain surrounding facilities where arrays of samplers were posted downwind of buildings or the land application site may have created turbulent air movement that diluted samples such that concentrations of PRRSV genomic material capture on filters were too low to produce a positive result by PCR.
Table 1. Cycle Threshold (Ct) values for sampling event 1
| Sample Description | Ct (Result) |
| Pen Floor, Room 17 | 37.5 (Suspect) |
| Fan Louver, Room 17 | 30.1 (Positive) |
| Feeder, Room 17 | 31.6 (Positive) |
| Air Filter, Room 17 | 31.2 (Positive) |
| Pen Floor, Room 18 | 31.5 (Positive) |
| Fan Louver, Room 18 | 31.4 (Positive) |
| Feeder, Room 18 | 37.6 (Suspect) |
| Air Filter, Room 18 | 30.5 (Positive) |
| Fecal Sample 1 | ³40 (Negative) |
| Fecal Sample 2 | ³40 (Negative) |
Cycle threshold (Ct) indicates the number of PCR cycles required for the sample fluorescence to reach a predefined threshold for identification (<38 = positive, ~38-40 = suspect, ≥40 = negative). Lower Ct values correspond to higher viral RNA concentration.
Table 2. Cycle Threshold (Ct) values for sampling event 2
| Sample Description | Ct (Result) |
| Exhaust Air, Room 5, 5 ft from fan | 33.1 (Positive) |
| Exhaust Air, Room 5, 12 ft. from fan | 34.1 (Positive) |
| Exhaust Air, Room 5, 19 ft. from fan | 38.1 (Suspect) |
| Indoor Air, Room 5, Rep 1 | 30.9 (Positive) |
| Indoor Air Room 5, Rep 2 | 33.3 (Positive) |
| Exhaust Air, Room 6, 5 ft from fan | 32.6 (Positive) |
| Exhaust Air, Room 6, 13 ft. from fan | 32.4 (Positive) |
| Flies | 37.0 (Suspect) |
| Flies Sterilized in Ethanol | 36.3 (Positive) |
| Maggots | 39.9 (Suspect) |
| Floor, Room 5, Rep 1 | 32.4 (Positive) |
| Floor, Room 5, Rep 2 | 32.3 (Positive) |
| Louvers, Room 5, Rep 1 | 33.1 (Positive) |
| Louvers, Room 5, Rep 2 | 32.1 (Positive) |
| Pens, Room 5, Rep 1 | 37.9 (Positive) |
| Pens, Room 5, Rep 2 | 35.8 (Positive) |
| Feeder, Room 5, Rep 1 | 35.8 (Positive) |
| Feeder, Room 5, Rep 2 | 37.5 (Suspect) |
| Pens, Room 4, Rep 1 | 35.6 (Positive) |
| Pens, Room 4, Rep 2 | 35.3 (Positive) |
| Floor, Room 4, Rep 1 | 31.4 (Positive) |
| Floor, Room 4, Rep 2 | 32.9 (Positive) |
| Louvers, Room 4, Rep 1 | 33.0 (Positive) |
| Louvers, Room 4, Rep 2 | 32.1 (Positive) |
Cycle threshold (Ct) indicates the number of PCR cycles required for the sample fluorescence to reach a predefined threshold for identification (<38 = positive, ~38-40 = suspect, ≥40 = negative). Lower Ct values correspond to higher viral RNA concentration.
Table 3. Cycle Threshold (Ct) values for sampling events 3 through 5
| Sampling Event | Sample Description | Ct (Result) |
| Event 3 | Air Filters (n=2) | ³40 (Negative) |
| Event 4 | Air Filters (n=4) | ³40 (Negative) |
| Fans (n=4) | ³40 (Negative) | |
| Oral Fluids, Room 15 | 34.0 (Positive) | |
| Oral Fluids, Room 16 | 36.1 (Positive) | |
| Oral Fluids, Room 17 | 38.0 (Suspect) | |
| Oral Fluids, Room 18 | 34.7 (Positive | |
| Event 5 | Air Filters (n=4) | ³40 (Negative) |
Cycle threshold (Ct) indicates the number of PCR cycles required for the sample fluorescence to reach a predefined threshold for identification (<38 = positive, ~38-40 = suspect, ≥40 = negative). Lower Ct values correspond to higher viral RNA concentration.
Future Plans
It is essential to identify which ambient weather conditions, if any, are favorable for air dispersion of infective PRRSv and which conditions will significantly limit dispersion. As research continues, the suspected ideal conditions for sampling downwind of mechanically ventilated PRRSv-positive barns or irrigation systems applying lagoon effluent from PRRSv-positive pigs will be 0 to 50°F with low to moderate wind speed and full cloud cover. At least 24 hours of continuous sampling is also expected to produce greater opportunity for positive air samples.
The continued inability to isolate the virus from manure samples is curious, given the universally positive samples we identified from the positive barns. However, the PRRSV is believed to require as few as 10 viral particles to be transmitted. Given the potentially very low concentration of viral material in manure, and the significant PCR inhibitors present in complex organic samples, the team continues to explore new sample preparation and testing methods for this matrix.
Lastly, further investigation into the potential roles of flies and maggots is warranted, particularly with the discovery of sufficient PRRSV genomic material in the gut of surface sterilized flies to yield a positive PRRSV result via RT-qPCR.
Authors
Presenting author
Logan Hafer, Undergraduate Research Assistant, Department of Biological Systems Engineering, University of Nebraska-Lincoln
Corresponding author
Dr. Amy Millmier Schmidt, Professor, Department of Biological Systems Engineering and Department of Animal Science, University of Nebraska-Lincoln, aschmidt@unl.edu
Additional author(s)
Dr. Benny Mote, Associate Professor, Department of Animal Science, University of Nebraska-Lincoln
Dr. Hiep Vu, Associate Professor, Department of Animal Science, University of Nebraska-Lincoln
Additional Information
-
- Porcine Reproductive and Respiratory Syndrome virus (PRRSV). Iowa State University – College of Veterinary Medicine; 2024 [accessed 2024 November 22]. https://vetmed.iastate.edu/vdpam/FSVD/swine/index-diseases/porcine-reproductive.
- Butler, J. E., Lager, K. M., Golde, W., Faaberg, K. S., Sinkora, M., Loving, C., & Zhang, Y. I. 2014. Porcine reproductive and respiratory syndrome (PRRS): an immune dysregulatory pandemic. Immunologic research, 59, 81-108. https://link.springer.com/article/10.1007/s12026-014-8549-5.
- Dee, S., T. Clement, and E. Nelson. 2023. Transmission of porcine reproductive and respiratory syndrome virus in domestic pigs via oral ingestion of feed material. J of the Am Vet Med Assoc, 262(1). https://doi.org/10.2460/javma.23.08.0447
- Osemeke, O.H., T. Donovan, K. Dion, D.J. Holtkamp and D.C.L. Linhares. 2021. Characterization of changes in productivity parameters as breeding herds transitioned through the 2021 PRRSV Breeding Herd Classification System. J Swine Health Prod. 2022;30(3):145-148. https://doi.org/10.54846/jshap/1269
Acknowledgements
Funding for this research was provided by the Nebraska Pork Producers Association under award #22-063 and an Undergraduate Student Research Program award from the UNL Institute of Agriculture and Natural Resources, Agricultural Research Division.
The authors are solely responsible for the content of these proceedings. The technical information does not necessarily reflect the official position of the sponsoring agencies or institutions represented by planning committee members, and inclusion and distribution herein does not constitute an endorsement of views expressed by the same. Printed materials included herein are not refereed publications. Citations should appear as follows. EXAMPLE: Authors. 2025. Title of presentation. Waste to Worth. Boise, ID. April 7–11, 2025. URL of this page. Accessed on: today’s date.
Call for Abstracts for Waste-to-Worth 2025
You are invited to participate!
The Waste to Worth Conference will be April 7-11, 2025 at the Grove Hotel in Boise, Idaho.
Waste to Worth 2025 welcomes oral, poster, panel, and workshop presentation proposals focused on applied solutions related to animal manure management and protecting the environment.
-
- Submissions should align with one or more of the general areas of emphasis (see below).
- Graduate students are encouraged to submit and participate in a poster presentation competition.
To submit an abstract, go to https://tinyurl.com/W2W2025
For more information, go to: https://wastetoworth.org or the W2W Call for Abstracts flyer.
Deadline is October 30, 2024
Areas of Emphasis
-
- Circular Bioeconomy
- Biosecurity
- Feed & Nutrient Management
- Manure, Soil Health & Sustainability
- Emerging Contaminants
- Manure Storage, Treatment, Handling & Application Systems
- Robotics & Artificial Intelligence in Animal Production Systems
- Value-Added Products from Agricultural Production Systems
- Climate Impacts & Adaptation/Mitigation Measures
- Air Quality, Emissions & Fate
- Educational Programming & Delivery
- Environmental Planning & Regulations in Animal Agriculture
- Case Studies/On-Farm Experience
- Mortality Management
- Sustainable Animal Systems
- New & Innovative Technology
- Water Quality
Who attends?
-
- Extension agents & specialists
- Progressive farmers & producers
- NRCS staff
- Consultants & technical experts
- Regulatory & policy advisors
- Scientists
- Technology providers
Implications of Manure Additives: Both Purposeful and Accidental
This webinar discusses purposeful additives like nitrification inhibitors and biochar as well as accidental additives like copper sulfate from disinfecting foot baths and how these things can and should impact our decisions when applying manure. This presentation was originally broadcast on September 22, 2023. Continue reading “Implications of Manure Additives: Both Purposeful and Accidental”
Moving Manure and Mortalities after Highly Pathogenic Avian Influenza
This webinar shares research and guidance on minimizing the risk of virus movement through manure and mortality management. This webinar also explains the roles technical advisors can play in response to an outbreak. This presentation originally broadcast on March 17, 2023. Continue reading “Moving Manure and Mortalities after Highly Pathogenic Avian Influenza”
Carbon Markets for Livestock Operations: Manure Treatment and Handling
The first in a series of 3 webinars, this presentation introduces the fundamentals of carbon emissions, as well as technologies, practices and market opportunities available to agricultural producers are critical to that transition on the livestock operation. This presentation was originally broadcast on November 18, 2022. Continue reading “Carbon Markets for Livestock Operations: Manure Treatment and Handling”
Worker Safety in Animal Production Systems
There’s more to worker safety than just bumps and bruises. This webinar discusses on-farm injuries related to manure and mortality handling and application as well as potential toxic gas exposures and how to minimize risks of each. This presentation was originally broadcast on October 21, 2022. Continue reading “Worker Safety in Animal Production Systems”

