Can Near-Infrared Reflectance Spectroscopy (NIRS) Be Used For Analyzing Horse Manure?
Increased horse numbers and insufficient acreage limit the ability for on-farm use of horse manure. Nearly 58% of surveyed farmers in NJ indicate that some manure was disposed off-farm while only 54% spread any manure on the farm (Westendorf et al., 2010). Analysis of manure by Near-Infrared Reflectance Spectroscopy (NIRS) could be a useful means of determining nutrient and energy content without time consuming efforts of wet chemistry and other laboratory analyses if horse manure is used as a fertilizer or energy source. The NIRS analysis works by subjecting samples to a concentrated light of a known spectrum and measuring the absorbance of the reflected beam (Dyer and Feng, 1997). Covalent chemical bonds of the common organic elements (Carbon, Nitrogen, Oxygen and Hydrogen) have strong absorbance in the NIRS region, useful because there is a correlation between absorbance and concentration (Malley et al., 2002). By comparing data between samples generated by NIRS to laboratory analysis of the same samples, NIRS equipment can be calibrated for practical use. The objectives of this project were: 1) determine the nutrient content and value of horse manure, and make NIRS calibrations based on previously determined wet chemistry values, and 2) determine if ash or Neutral Detergent Fiber (NDF) content can be used to predict Gross Energy (GE) levels.
What did we do?
Horse manure consisting of 123 solid dry stack manure samples, were collected from 30 NJ farms over four seasons during a 12-month period in 2008-2009. Samples were collected from various random locations in a manure pile in ~ 4 l sealable plastic bags, frozen, and stored until analysis. All samples were dried at 55o C to a constant weight in a Thermocore® oven. Following drying, all samples were ground to a particle size of 5-10 mm in a Waring® industrial blender, referred to as Coarse ground samples. Samples were sent to DairyOne Laboratories in Ithaca, NY and analyzed for manure components (Total-N, P2O5, K2O, NDF, and GE); samples were analyzed for Ash by the Rutgers University Soil Testing Laboratory. Coarse ground samples were further ground in a coffee grinder to a particle size between 2-3 mm (these samples are referred to as Fine ground samples). All NIRS analysis of Coarse and Fine ground samples were made with a Unit y Scientific Spectrastar ™ 2400 Drawer model (Brookfield, CT). Samples were scanned at 1nm intervals over the wavelength range of 1250-2350 nm, as prescribed by Unity Scientific. Data from the DairyOne Laboratory results were used as reference values to develop calibrations using the Ucal™ software package (Unity Scientific, Brookfield, CT) set at default values using a partial linear squares statistical model.
What have we learned?
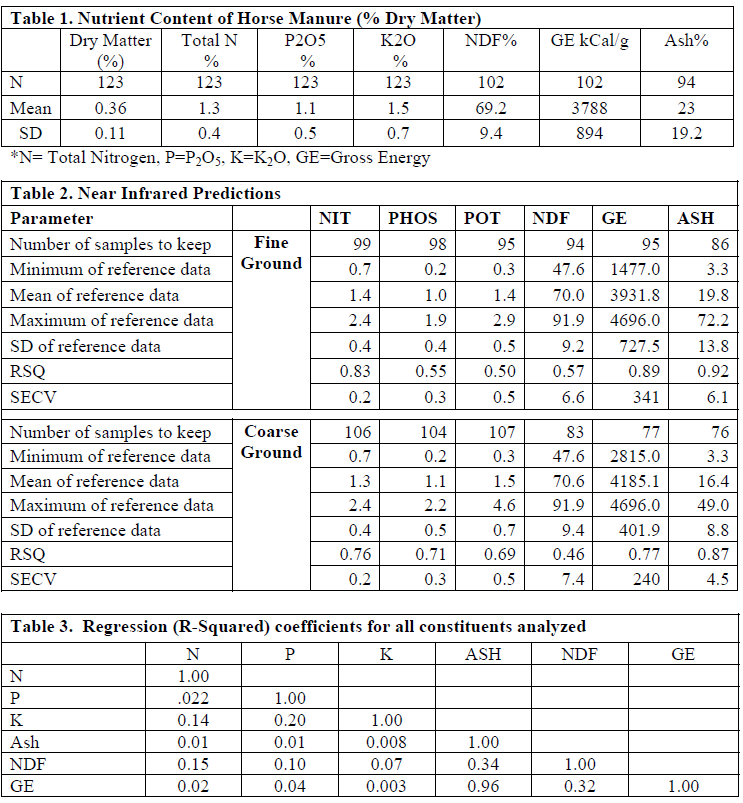
On a dry matter basis (Table 1) samples averaged 1.3% N, 1.1% P2O5, 1.5% K2O, 69.2% NDF, 3800 kCal/g GE, and 24% Ash. The NIRS equations (Table 2) for Coarse (5-10 mm) ground horse manure predicted nutrient content, R-squared values of 0.76, 0.71, 0.69, 0.46, 0.77, and 0.87 for N, P2O5, K2O, NDF, GE, and Ash, respectively. The NIRS also predicted Fine (2-4 mm) ground horse manure R-squared values of 0.83, 0.55, 0.50, 0.57, 0.89, and 0.92 for N, P2O5, K2O, NDF, GE, and Ash, respectively. Ash, GE and NDF were regressed to determine how effectively Ash and NDF would predict GE (Table 3); NDF was a poor predictor of GE content (R-squared of 0.32), while Ash was a good predictor (R-squared of 0.96).
Future Plans
This research suggests that NIRS can be useful for predicting nutrient content of horse manure and that Ash is a good predictor of energy content. A comparative field trial on horse farms is planned for follow-up.
Authors
Michael L. Westendorf. Extension Specialist in Animal Science. Rutgers, the State University of New Jersey westendorf@aesop.rutgers.edu
Zane R. Helsel. Extension Specialist in Plant Biology and Pathology. Rutgers, the State University of New Jersey.
Additional information
Author Contact Information:
Michael Westendorf
Rutgers, The State University of New Jersey
84 Lipman Drive
New Brunswick, NJ 08901
Phone: 848-932-9408
e-mail: westendorf@aesop.rutgers.edu
Reference:
Dyer, D. J. and P. Feng. 1997. NIR Destined to be Major Analytical Influence. Feedstuffs Magazine. November 10, 1997.
Malley, D.F., Yesmin, L., and Eilers, R. G. 2002. Rapid Analysis of Hog Manure and Manure- amended Soils Using Near-infrared Spectroscopy. Soil Science Society of America Journal. 2002. 1677-1686.
Westendorf, M. L., T. Joshua, S. J. Komar, C. Williams, and R. Govindasamy. 2010. Manure Management Practices on New Jersey Equine Farms. Prof. Anim. Sci. 26:123-129.
Acknowledgements
Supported in part by the State Equine Initiative. Rutgers Equine Science Center. New Jersey Agricultural Experiment Station.
The authors are solely responsible for the content of these proceedings. The technical information does not necessarily reflect the official position of the sponsoring agencies or institutions represented by planning committee members, and inclusion and distribution herein does not constitute an endorsement of views expressed by the same. Printed materials included herein are not refereed publications. Citations should appear as follows. EXAMPLE: Authors. 2015. Title of presentation. Waste to Worth: Spreading Science and Solutions. Seattle, WA. March 31-April 3, 2015. URL of this page. Accessed on: today’s date.


 What did we do?
What did we do?


 What did we do?
What did we do?