Most people alive today have never known a world without antibiotics. Antibiotics became widely used to treat bacterial infections in the 1930s and ‘40s, and were quickly labeled a “miracle drug”. But, as the widespread use of antibiotics evolved, so did the discovery of antibiotic-resistant bacteria. And we have now entered an era where antibiotic resistance is considered one of the biggest worldwide public health challenges of our time.
What is antibiotic/antimicrobial resistance?
When a microorganism (e.g. bacterium, virus, etc.) adapts in some way to resist the effect of a medicine or product used to kill it, it is said to have developed “antimicrobial resistance,” or AMR. Bacteria are one kind of microorganism – others are viruses, molds, fungi, and yeasts. Antibiotic resistance (ABR) refers to the ability of bacteria to resist the effects of one or more antibiotics. An example of antibiotic resistance is methicillin-resistant Staphylococcus aureus, or MRSA, which is a type of bacteria that can cause a skin infection that is difficult to treat.
AMR is not something new, this phenomenon has been occurring in the environment as a natural phenomenon since thousands of years ago, where microorganisms have been competing for space and resources. As a result of that, some microorganisms have evolved to produce antibiotics (penicillin was discovered from soil bacteria!) and other microorganisms have created methods to be resistant to those substances. AMR genes that give bacteria protection against antibiotics have been found in 30,000 years old DNA coming from Beringian permafrost sediments!
AMR has not always been linked to human use of antibiotics, but the modern use of those substances in human medicine and food animal production has accelerated the creation of new AMR for drugs that are threatening human health. The use of antibiotics on food animal production is only one of the causes of AMR. Human medicine has a big role in this fight, and ANY use of antibiotics can produce AMR. Antibiotics can also be used as anaphylaxis, which occurs when they are used as a preventive measure (i.e. before surgeries, patients are prescribed antibiotics in an attempt to prevent any infection that could result from the surgical procedure). Similar to the use as a treatment against diseases, the preventive use of antibiotics represents an AMR risk.
As we can see, AMR is a complex topic. As antibiotics are part of our life, everyone has a role in the mitigation of this human health threat!
Antimicrobials and food animals:

Antimicrobial resistance can reach humans in different ways: from people to people, animals to people, environment to people; and we are still learning and discovering new spread ways. When those AMR microorganisms are pathogenic, they can result in diseases difficult or impossible to treat in humans. AMR is among the top 10 global public health threats, according to the World Health Organization.
Similar to the use of antibiotics in humans, food animal production utilizes antibiotics to treat and prevent diseases and, in some species, to promote growth. Those animals can be a reservoir for AMR, especially when antibiotics are overused or misused. Normally, those bacteria are present in the animal’s gut microbiome and are expelled to the environment via excreted manure. Understanding the relation between the use of antimicrobials in food humans and its impacts on the transmission of AMR to humans is a key point in the fight against AMR.
Among the antimicrobials used in food animal production are those deemed “medically important” by the Food and Drug Administration (FDA). Those termed “medically important” are drugs that are used for treating human disease and animal disease. Many drugs used in veterinary medicine are not used in human medicine.
Cattle and swine were, in 2018, the animals that had the largest consumption of medically important drugs, representing 42% and 39% of the 6,653 tons of antimicrobials sold in that year in the US. In the same year, food animals consumed 6,096 tons of non-medically important antimicrobial drugs, with cattle and chicken consuming the largest amounts (61% and 25%, respectively). Tetracyclines (66%), penicillins (12%), and macrolides (8%) were the most prevalent medically important antimicrobial families used in food animals in that year (FDA, 2019).
The transition of AMR from animals to humans:
Similar to the usage of antimicrobials in human health, their use in food animal production creates conditions for the multiplication of AMR microorganisms. When those organisms are resistant to medically important drugs and infect humans, they became a human health problem. In the US, AMR is responsible for 2.8 million infections and 35,000 deaths per year (CDC, 2019).
Links between food animals and common foodborne pathogens like E. coli, Salmonella, enterococci, and Staphylococcus aureus show the important role of antibiotic consumption in animals. AMR bacteria can pass AMR characteristics to their descendants, that process is called vertical gene transfer. Also, bacteria can achieve AMR properties through horizontal gene transfer (HGT), a process where microorganisms exchange genetic elements containing AMR genes. HGT is one mechanism by which bacteria adapt to resist the impacts of antibiotics used to treat them.
Several studies have confirmed the transfer of AMR bacteria from animals to humans. This process begins when AMR bacteria spreads inside of food animals after long-term use of antibiotics. Antibiotics kill susceptible bacteria and facilitate the growth of AMR bacteria. AMR bacteria can reach the environment after being excreted, and then, via food or contaminated material, reach the human body. Several AMR bacteria affecting humans have been tracked to an animal or food source worldwide.
AMR Prevention Strategies
Several controls on antimicrobial use in food animals have been imposed as a measure to decrease the spread of AMR (Table 1). These bans target antimicrobial growth promoters (AGP), however, the control of antimicrobials in food production must balance the concerns over growing AMR and the positive impacts on animal health, and welfare.
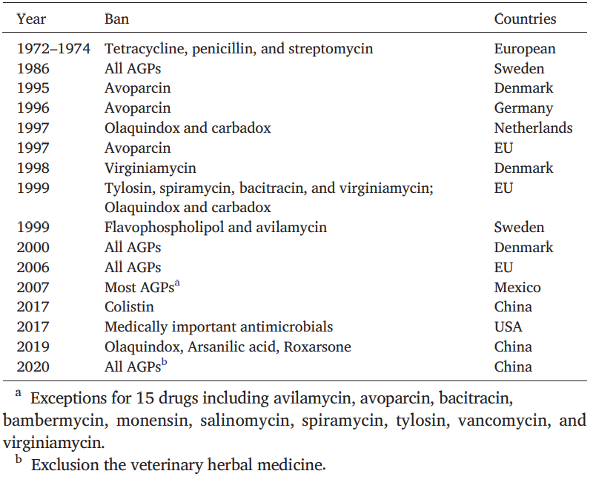 Table 1. Timeline for the legal ban of Antimicrobial growth promoters in food animal production. Developed by Ma et al., 2020.
Table 1. Timeline for the legal ban of Antimicrobial growth promoters in food animal production. Developed by Ma et al., 2020.Farmers can reduce their risk of AMR in their operations by adopting practices recommended by the CDC in animal production:
- Work closely with your veterinarian. Good communication with your veterinarian will help you to decide when antibiotics are needed and what specific actions you can take in your operation to prevent AMR infections.
- Keep animals healthy. Healthy animals are less likely to become sick and require antimicrobials! Follow good husbandry and biosecurity practices, care about your animal welfare standards, vaccinate according to your veterinarian recommendations, and implement an appropriate waste management plan.
- Use antibiotics exactly as prescribed. Overuse and misuse of antibiotics in human health and animal production could help the dissemination of AMRs. When using antibiotics in your operation, always use the “dose (amount), duration (period of time), and route of administration (how to give antibiotics)” as described by your veterinarian. Keep records of all your antibiotics usage and safely dispose of any extra antibiotics.
- Protect yourself and your workers. Make sure that you and your workers/coworkers have an appropriate vaccination, safety training, personal protective equipment, toilets, and handwashing stations.
We all have a role in managing the spread of AMR.
Authors
Juan Carlos Ramos Tanchez – – written while a student at the University of Nebraska as a part of a course in animal manure management and originally published on water.unl.edu/manure
This article was reviewed by Amy Schmidt, Nebraska Extension Specialist, and Randy Saner, Nebraska Extension Educator
Further Reading
i(AM)Responsible project’s Twitter and Facebook accounts.
FDA’s Strategy on Antimicrobial Resistance – Questions and Answers.
CDC. (2019). Antibiotic Resistance Threats in the United States, 2019. Atlanta, GA: U.S. Department of Health and Human Services, CDC.
FDA. (2019). 2018 Summary Report on Antimicrobials Sold or Distributed for Use in Food-Producing Animals. U.S. Food and Drug Administration.
Ma, F., Xu, S., Tang, Z., Li, Z., & Zhang, L. (2020). Use of antimicrobials in food animals and impact of transmission of antimicrobial resistance on humans. Biosafety and Health, 1-7. doi:https://doi.org/10.1016/j.bsheal.2020.09.004.

