Links to PEDv (Porcine Epidemic Diarrhea Virus).
Microorganisms
Microorganisms (e.g. virus, bacteria, protozoa, and fungi) surround us, on us, and in us; they are ubiquitous and everything in the world is governed by them. They are part of our everyday lives. They influence the the quality of our soil, food grown on that soil, and how our body reacts to that food. They are diverse, ranging from a simple mix of protein and DNA to complex multi-cellular small “animals”. Most environmental microorganisms spend their entire lives as quiet members of their ecological society, but some reach a level of infamy. Pathogens may only represent a very small portion of all microorganisms, but they are often the most visible, thanks to readily reported outbreaks, food recalls, and proliferation of internet news blogs and sites.
What is a Pathogen?
A pathogen is a biological agent that causes disease or illness; this disease can occur in humans, animals, or crops. Zoonotic pathogens refers to pathogens naturally transmitted from animals to humans and are often heard about on news sites or involved in food recalls.
All animals including pets, livestock, wildlife and humans, are possible hosts of potential human pathogens. We will focus on pathogens originating from livestock and poultry that might be transported to humans via air, water, soil, crop, and fomites (inanimate objects) contacted directly or indirectly by manure.
Zoonotic Pathogens
There are four general classes of zoonotic pathogens:
- viruses
- bacteria
- protozoan parasites
- helminth parasites
Zoonotic viruses are those found mainly in animals that cause disease in people who come into contact with the animal or share a vector (transmitter of disease) like a mosquito (West Nile Virus is a virus of birds which mosquitoes carry and can transmit to people). Viruses can only multiply when they are inside a host cell.
- Until very recently, it was considered that most fecal or urine transmitted viruses of livestock were not zoonotic, but things have changed somewhat in recent years, and we are now in a steep learning curve as to how important ruminants and poultry are as reservoirs of these zoonotic viral agents.
Zoonotic bacterial pathogens are, like all bacteria, single celled microorganisms that can survive and, under favorable conditions, reproduce in terrestrial and aquatic environments. The zoonotic bacteria are those that typically cycle in domestic animals without causing disease in their typical hosts. However, when they get transmitted into people, the disease that is produced can be severe.
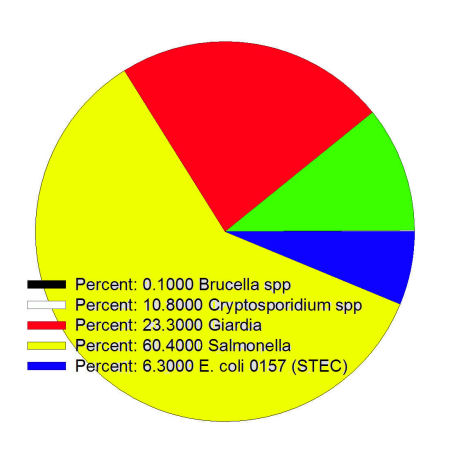
- Examples of zoonotic bacteria are Salmonella spp., strains of Escherichia coli such as E. coli 0157:H7, Listeria monocytogenes, and Campylobacter spp.
Zoonotic protozoan parasites, are protozoa that are found in other animals and which can infect people. There are basically two roles that humans can play in this scenario. They can be accidental hosts in the life cycle of the protozoan, where the protozoan undergoes the same development in the human as it does in its normal reservoir host. Or, the human may be an intermediate host in the life cycle of the parasites, just like any other vertebrate; in this case, the reservoir host shed many stages into the environment with the goal of infecting as many intermediate hosts as possible.
- In the case of zoonotic protozoa relative to domestic farm animals, only a few have proven to be of significant concern relative to the infection of people.
- Species of Cryptosporidium found in horses, cattle, pigs, and sheep can accidentally infect people, with C. parvum of young ruminants being the most common offender.
- Giardia of livestock typically does not seem to occur in people, but it does seem that they might be infected with the human form and could then serve as a source of stages that might be passed to humans.
Zoonotic helminth parasites are worms, nematodes (roundworms), cestodes (tapeworms), or trematodes (flukes), that have cycles similar to protozoa. Again, people can be infected accidentally by the worm in the same manner as a reservoir host or they can be serving as just another vertebrate intermediate host in the life cycle of the parasite.
- Fortunately, in the case of domestic farm animals, the helminth parasites are for the most part not zoonotic with respect to people. The only forms with stages that might be infectious to people from manure would be the egg of the pig roundworm, Ascaris suum.
Photo source: Jeanette Thurston-Enriquez webcast presentation.
Detailed discussion of protozoan parasites, bacteria, and viruses can be found on pages 5, 12, and 18, respectively, of the USDA NRCS technical note
Waterborne Pathogens in Agricultural Watersheds
Several outbreaks of human illness and death have been attributed to drinking water contaminated with livestock manure. Of 66 drinking water outbreaks in affluent nations, the probable cause of 12 of the outbreaks was livestock manure (see Hrudey and Hrudey, 2004 in Research Summaries. These included:
- An outbreak at the 1999 Washington County Fair, New York (E. coli O157:H7; of 781 confirmed cases, 71 people were hospitalized, and 2 died);
- An outbreak in Walkerton, Ontario, Canada in 2000 (E. coli O157:H7 and Campylobacter jejuni; 2,300 people were ill, 65 were hospitalized and 7 died).
These outbreaks were indicative of the capability of the pathogens to survive and be leached through soil to groundwater sources of drinking water.
Not all illness outbreaks are livestock related. For example, animal manure was initially suggested as the source of the largest drinking water outbreak in U.S. history – the Cryptosporidium outbreak in Milwaukee, WI in 1993. Several years later following advances in microbiology and genetics, human sewage was identified as the likely contributor.
Antibiotic Resistant Bacteria in Agricultural Manures
An antibiotic-resistant bacterial population is one in which resistance is either intrinsic or has been acquired from exposure either to antibiotics or to other antibiotic resistant bacterial populations. The increased frequency of antibiotic resistant pathogens has become a serious public health concern as demonstrated with outbreaks of methicillin-resistant Staphylococcus aureus (MRSA) and antibiotic resistant Salmonella such as Salmonella DT104. Little research and information is available on the presence of antibiotic resistant bacteria originating in manure and manure land applied environments, and, thus, little is known about their fate and transport in soil, water, crops, and agronomic systems.
A listing of possible zoonotic pathogens can be found on pages 6 – 10 of an EPA literature review.
Authored by: Michael Jenkins and John Brooks, USDA ARS, Dwight Bowman and Janice Liotta, Cornell University.